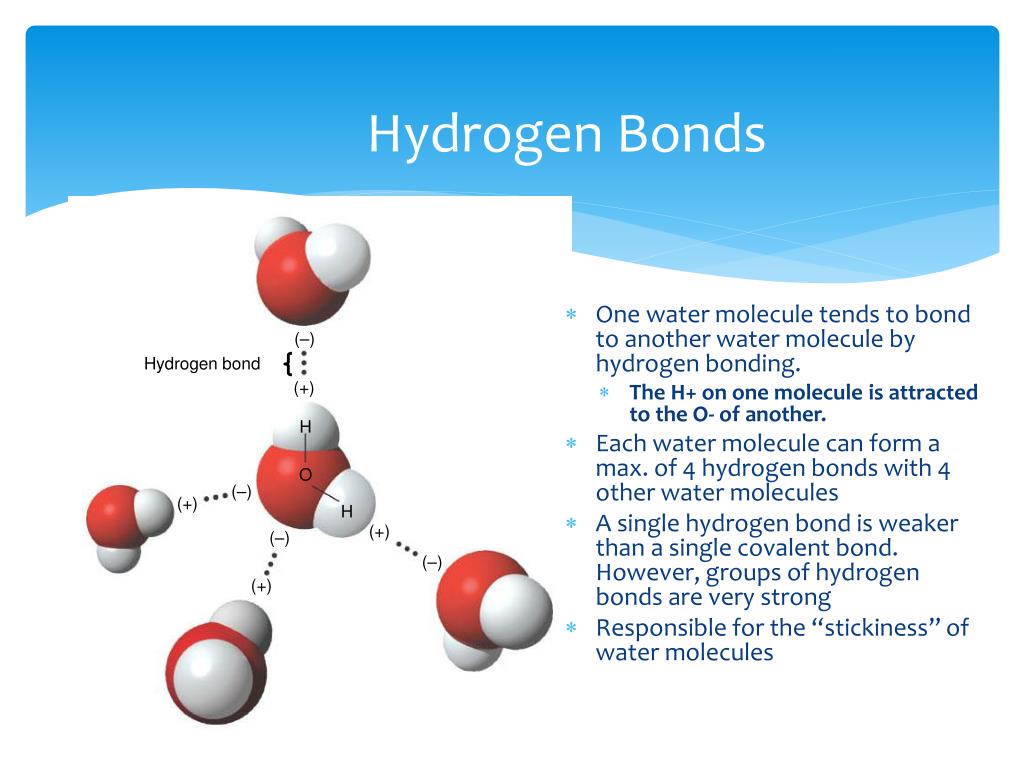
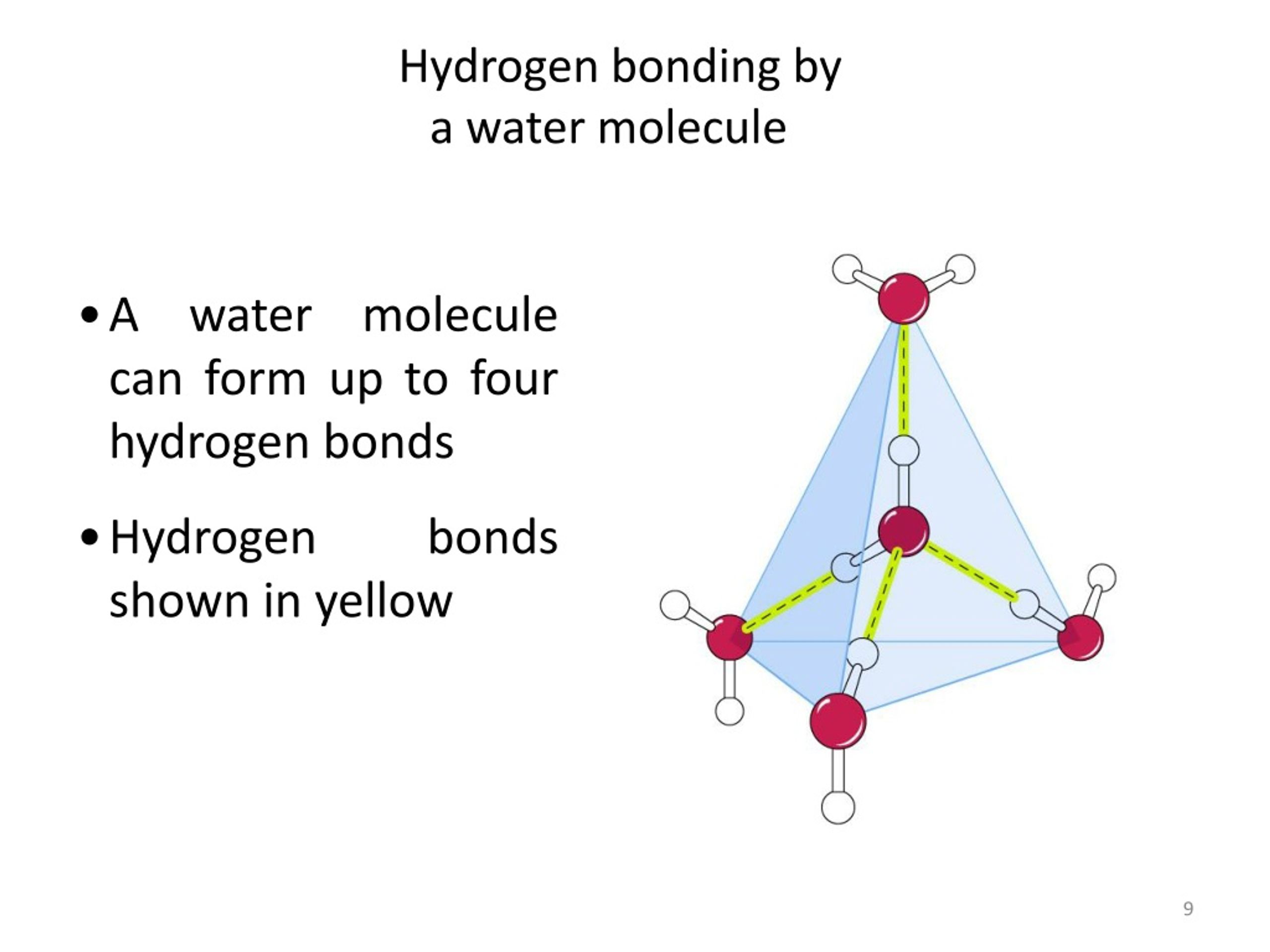
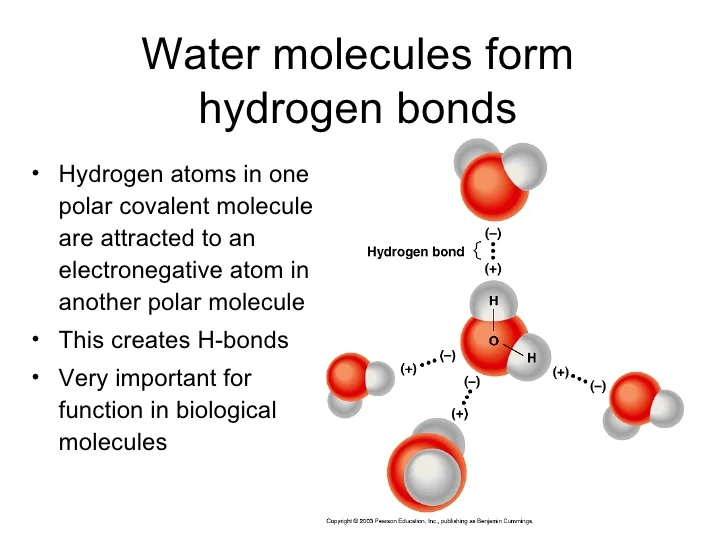
How Many Hydrogen Bonds Can One Water Molecule Form - While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. Positive hydrogen of one molecule. Another hydrogen bond can be formed using the other lone pair on the oxygen. Each oxygen atom in a water molecule has two lone pairs of. It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. How many hydrogen bonds can a single water molecule form? Explain hydrogen bonding in terms of water. The maximum number of hydrogen bonds that a water molecule can form is four. So far, we’ve drawn this water molecule with one hydrogen bond. A single water molecule can form up to 4 hydrogen bonds:
While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. The maximum number of hydrogen bonds that a water molecule can form is four. Positive hydrogen of one molecule. A single water molecule can form up to 4 hydrogen bonds: So far, we’ve drawn this water molecule with one hydrogen bond. Explain hydrogen bonding in terms of water. How many hydrogen bonds can a single water molecule form? Another hydrogen bond can be formed using the other lone pair on the oxygen. It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. Each oxygen atom in a water molecule has two lone pairs of.
Each oxygen atom in a water molecule has two lone pairs of. The maximum number of hydrogen bonds that a water molecule can form is four. How many hydrogen bonds can a single water molecule form? Explain hydrogen bonding in terms of water. So far, we’ve drawn this water molecule with one hydrogen bond. It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. Positive hydrogen of one molecule. Another hydrogen bond can be formed using the other lone pair on the oxygen. While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. A single water molecule can form up to 4 hydrogen bonds:
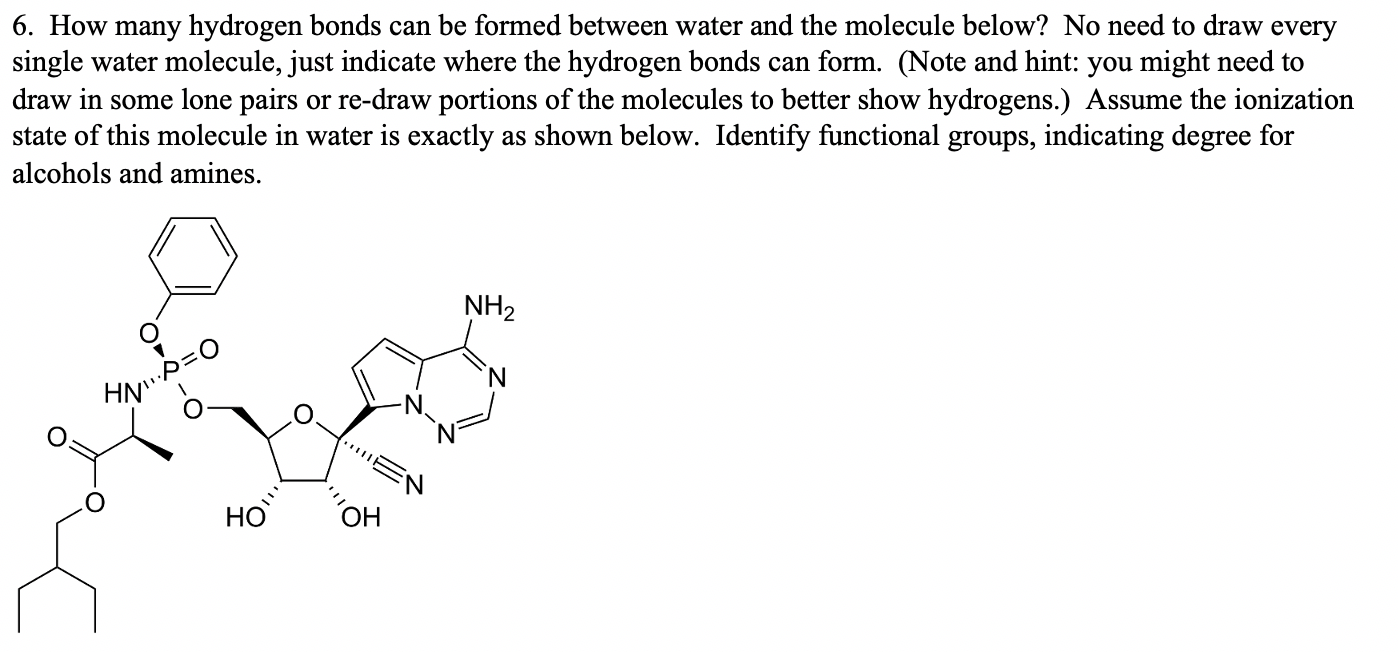
Solved 6. How many hydrogen bonds can be formed between
So far, we’ve drawn this water molecule with one hydrogen bond. Explain hydrogen bonding in terms of water. A single water molecule can form up to 4 hydrogen bonds: Positive hydrogen of one molecule. It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2.
PPT Properties of Water PowerPoint Presentation, free download ID
The maximum number of hydrogen bonds that a water molecule can form is four. Positive hydrogen of one molecule. Explain hydrogen bonding in terms of water. Another hydrogen bond can be formed using the other lone pair on the oxygen. While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities.
Unit 2 (Biochemistry) Notes, Part 1 Atomic And Molecular Structure
So far, we’ve drawn this water molecule with one hydrogen bond. While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. Positive hydrogen of one molecule. How many hydrogen bonds can a single water molecule form? Each oxygen atom in a water molecule has two lone pairs of.
Overview A Chemical Connection to Biology ppt download
While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. How many hydrogen bonds can a single water molecule form? A single water molecule can form up to 4 hydrogen bonds: Another hydrogen bond can be formed using the other lone pair on the oxygen. The maximum number of hydrogen bonds that a.
PPT Lec.1 Chemistry Of Water PowerPoint Presentation, free download
So far, we’ve drawn this water molecule with one hydrogen bond. Another hydrogen bond can be formed using the other lone pair on the oxygen. It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. Each oxygen atom in a water molecule has two lone pairs of. Explain hydrogen bonding in terms of water.
Hydrogen bonding
It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. Explain hydrogen bonding in terms of water. While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. A single water molecule can form up to 4 hydrogen bonds: How many hydrogen bonds can a single water molecule form?
Water Review
A single water molecule can form up to 4 hydrogen bonds: Explain hydrogen bonding in terms of water. While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities. So far, we’ve drawn this water molecule with one hydrogen bond. Each oxygen atom in a water molecule has two lone pairs of.
Showing a water molecule with Hydrogen bonds There are still two
A single water molecule can form up to 4 hydrogen bonds: Explain hydrogen bonding in terms of water. Positive hydrogen of one molecule. Another hydrogen bond can be formed using the other lone pair on the oxygen. The maximum number of hydrogen bonds that a water molecule can form is four.
Water Molecule Polarity And Hydrogen Bond Formation
How many hydrogen bonds can a single water molecule form? Each oxygen atom in a water molecule has two lone pairs of. Another hydrogen bond can be formed using the other lone pair on the oxygen. Explain hydrogen bonding in terms of water. Positive hydrogen of one molecule.
Diagram Og Hydrogen Bonds Between Water Molecule] 3.11 Bioc
How many hydrogen bonds can a single water molecule form? It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. Another hydrogen bond can be formed using the other lone pair on the oxygen. Each oxygen atom in a water molecule has two lone pairs of. The maximum number of hydrogen bonds that a water molecule.
So Far, We’ve Drawn This Water Molecule With One Hydrogen Bond.
It can donate 2 hydrogen bonds (via its two hydrogen atoms) and accept 2. Another hydrogen bond can be formed using the other lone pair on the oxygen. The maximum number of hydrogen bonds that a water molecule can form is four. A single water molecule can form up to 4 hydrogen bonds:
Each Oxygen Atom In A Water Molecule Has Two Lone Pairs Of.
Positive hydrogen of one molecule. How many hydrogen bonds can a single water molecule form? Explain hydrogen bonding in terms of water. While water can form up to four hydrogen bonds per molecule, other common molecules have different capacities.




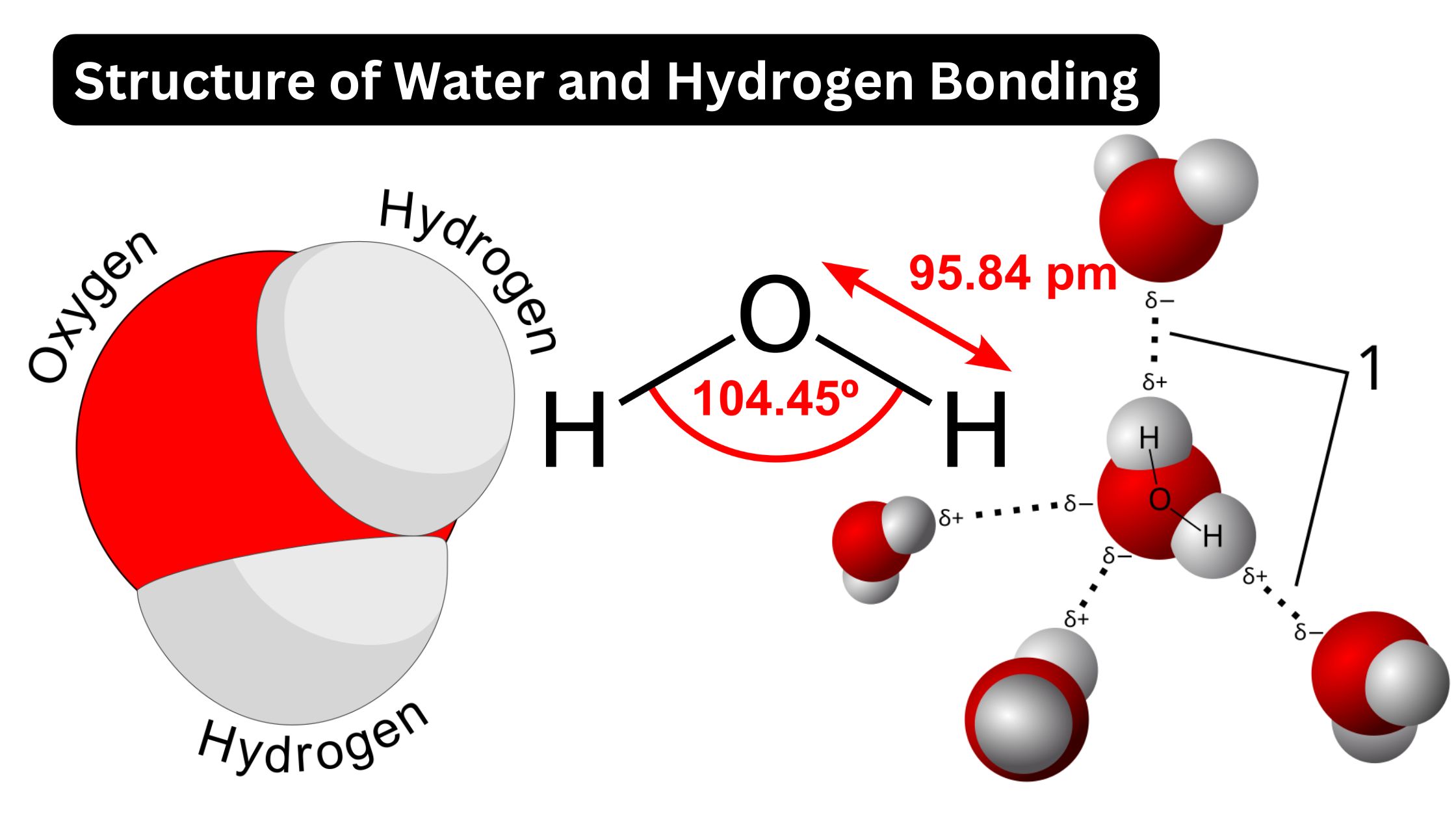
![Diagram Og Hydrogen Bonds Between Water Molecule] 3.11 Bioc](https://d20khd7ddkh5ls.cloudfront.net/hydrogen_bonding_5.png)