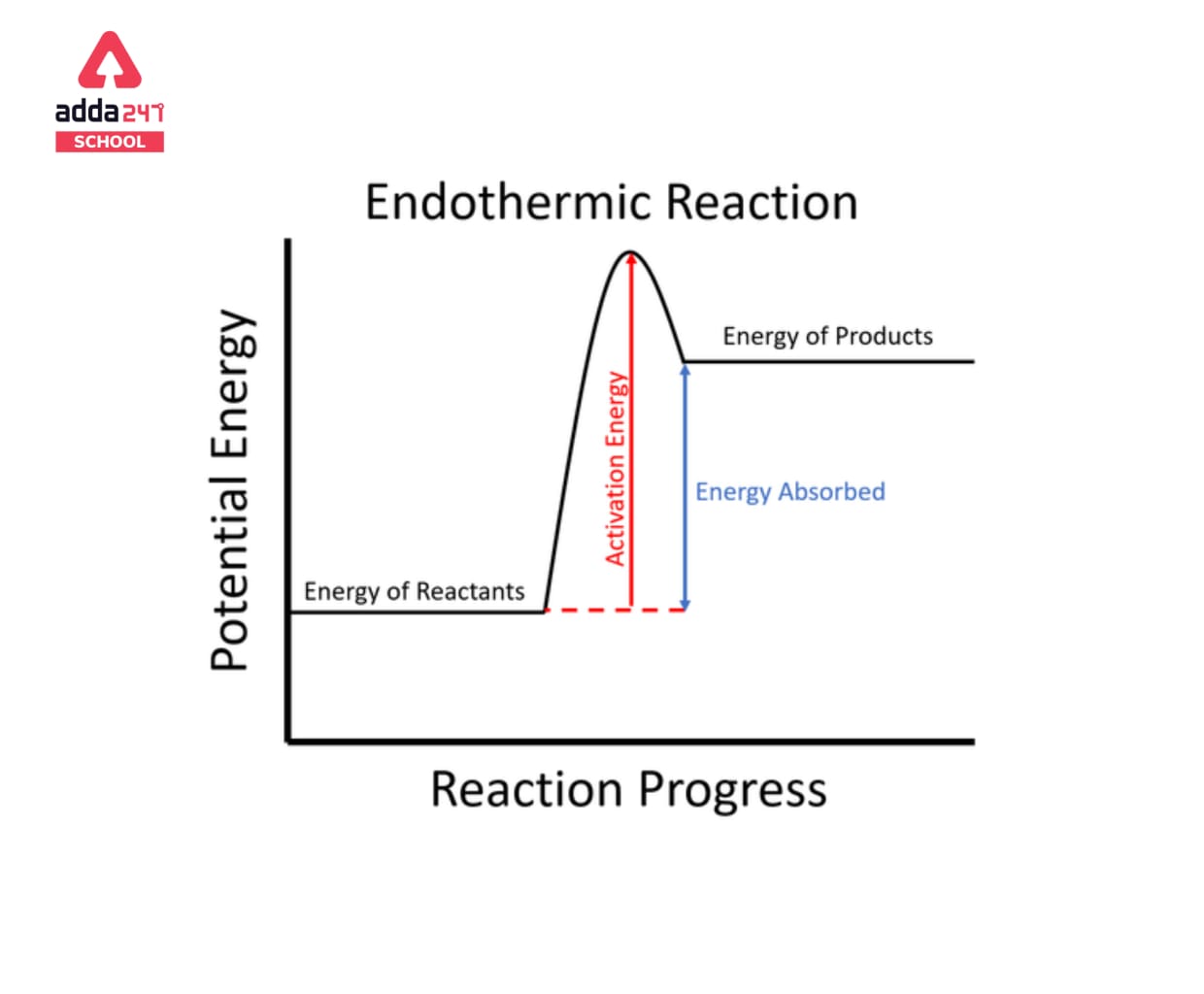
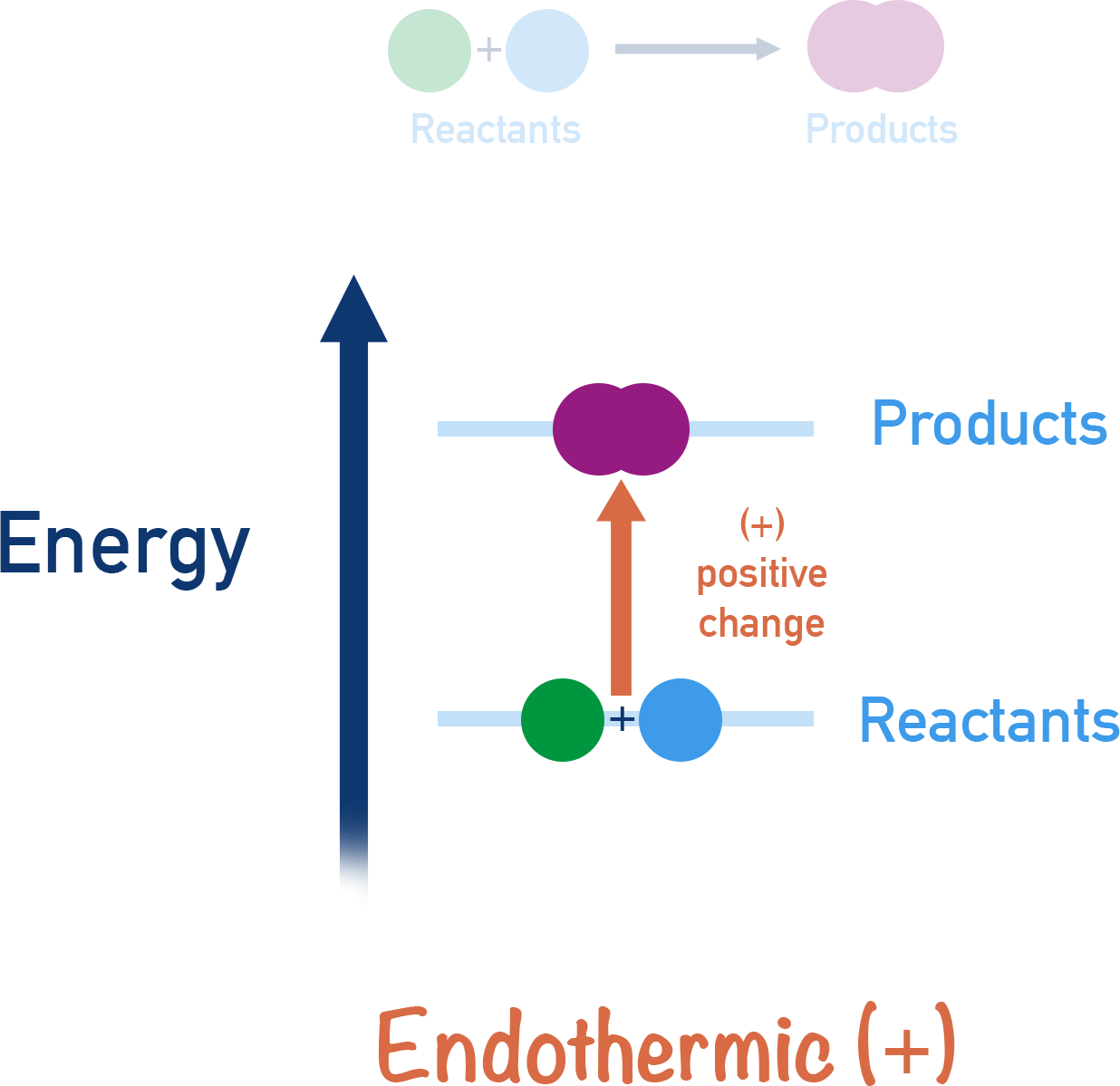
Which Form Of Energy Does An Endothermic Reaction Use - It results in a decrease in. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses energy primarily in the form of chemical energy to proceed. Chemical energy results from the particular arrangement of atoms in a chemical compound, and during an endothermic reaction,. This energy is absorbed from the surroundings,. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction uses chemical energy.
An endothermic reaction uses energy primarily in the form of chemical energy to proceed. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. In an endothermic reaction, energy is absorbed from the surroundings, causing the. Chemical energy results from the particular arrangement of atoms in a chemical compound, and during an endothermic reaction,. This energy is absorbed from the surroundings,. An endothermic reaction uses chemical energy. It results in a decrease in.
In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses chemical energy. Chemical energy results from the particular arrangement of atoms in a chemical compound, and during an endothermic reaction,. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. This energy is absorbed from the surroundings,. It results in a decrease in. An endothermic reaction uses energy primarily in the form of chemical energy to proceed.
Comparing Endothermic And Exothermic Potential Energy
An endothermic reaction uses chemical energy. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. It results in a decrease in. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses energy primarily in the form of chemical energy to proceed.
14.3 Energy and Reactions. ppt download
It results in a decrease in. This energy is absorbed from the surroundings,. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction uses energy primarily in the form of chemical energy to proceed. An endothermic reaction is a chemical reaction that uses thermal energy,.
Endothermic Reaction Examples with Equations
This energy is absorbed from the surroundings,. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction uses energy primarily in the form of chemical energy to proceed. It results in a decrease.
The Visual Representation of an Endothermic Reaction Enthalpy Diagram
It results in a decrease in. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. An endothermic reaction uses chemical energy. This energy is absorbed from the surroundings,. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system.
Enthalpy Changes (ALevel) ChemistryStudent
An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. This energy is absorbed from the surroundings,. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction.
Which Form Of Energy Does An Endothermic Reaction Use
Chemical energy results from the particular arrangement of atoms in a chemical compound, and during an endothermic reaction,. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. It results in a decrease in. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from.
Anatomy of an Endothermic Reaction Visualizing the Energy Diagram
An endothermic reaction uses chemical energy. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. It results in a decrease in. An endothermic reaction uses energy primarily in the form of.
Endothermic Reactions Definition and Examples
In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses energy primarily in the form of chemical energy to proceed. This energy is absorbed from the surroundings,. It results in a decrease in. An endothermic reaction uses chemical energy.
The Process of a TwoStep Endothermic Reaction
It results in a decrease in. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. An endothermic reaction uses energy primarily in the form of chemical energy to proceed. An endothermic reaction uses chemical energy.
Exploring the Energy Profile of Endothermic Reactions
This energy is absorbed from the surroundings,. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. An endothermic reaction.
An Endothermic Reaction Uses Chemical Energy.
An endothermic reaction is a chemical reaction that uses thermal energy, or heat, from its surroundings. In an endothermic reaction, energy is absorbed from the surroundings, causing the. An endothermic reaction uses energy primarily in the form of chemical energy to proceed. An endothermic reaction uses chemical energy to drive the process, which often comes from heat or electrical energy applied to the system.
Chemical Energy Results From The Particular Arrangement Of Atoms In A Chemical Compound, And During An Endothermic Reaction,.
This energy is absorbed from the surroundings,. It results in a decrease in.