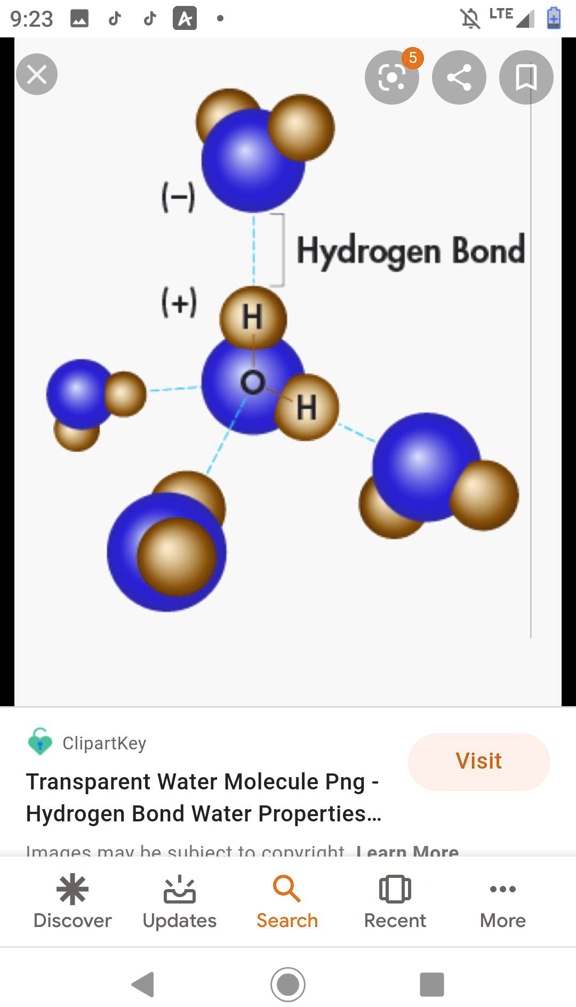
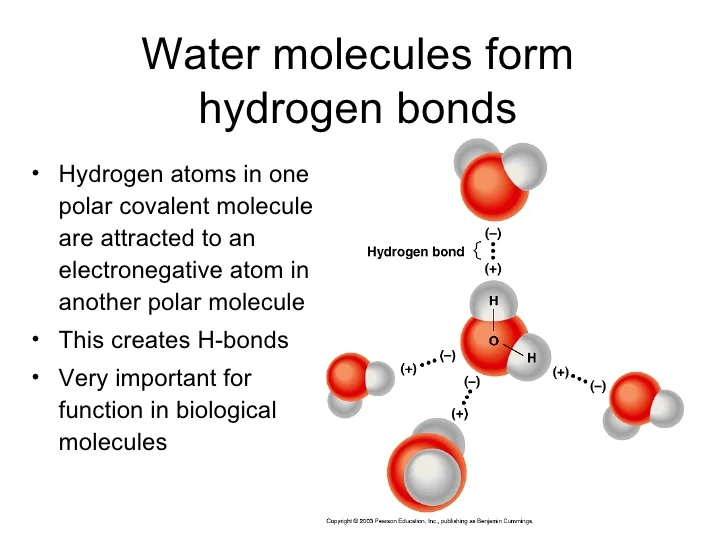
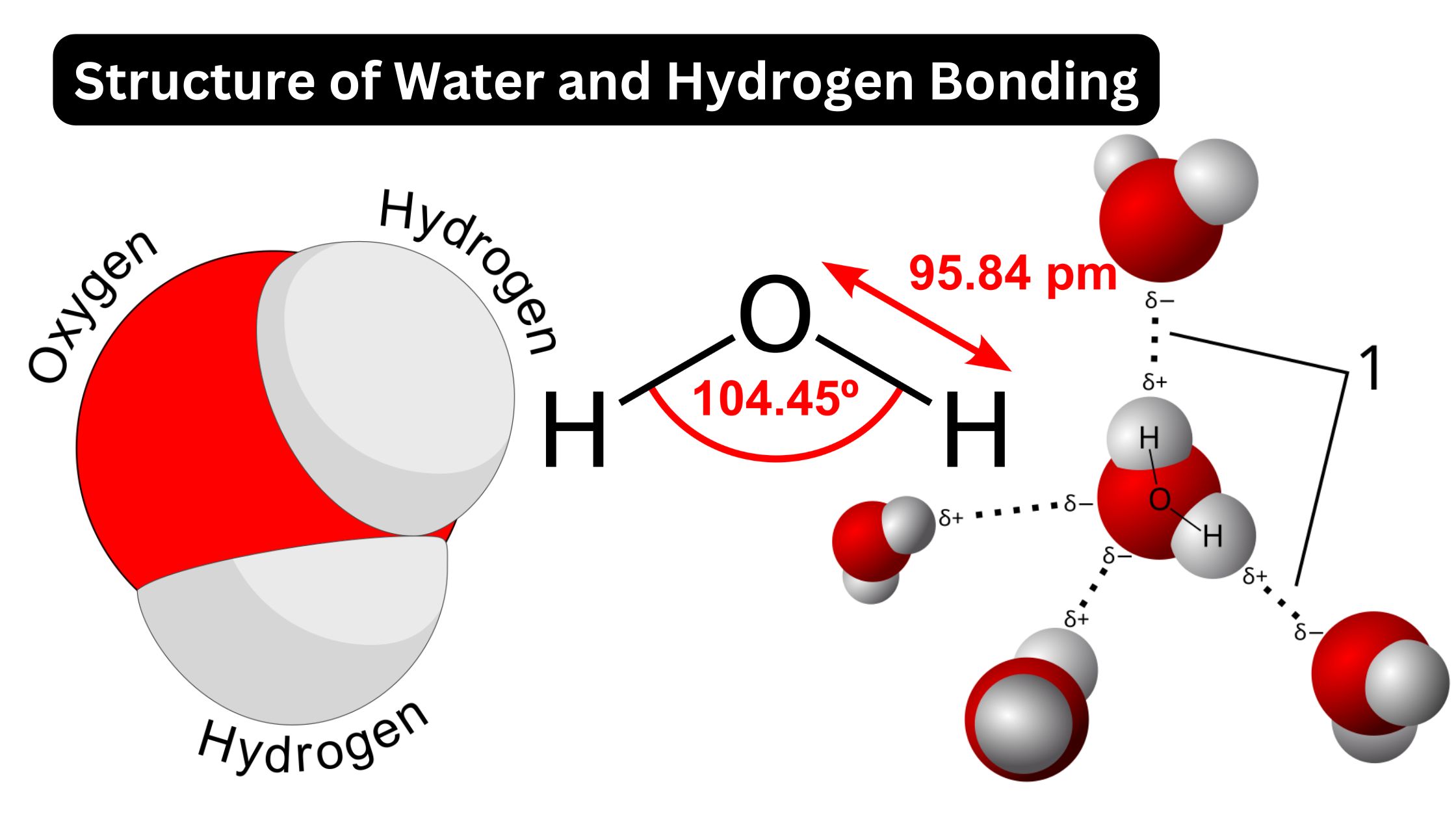
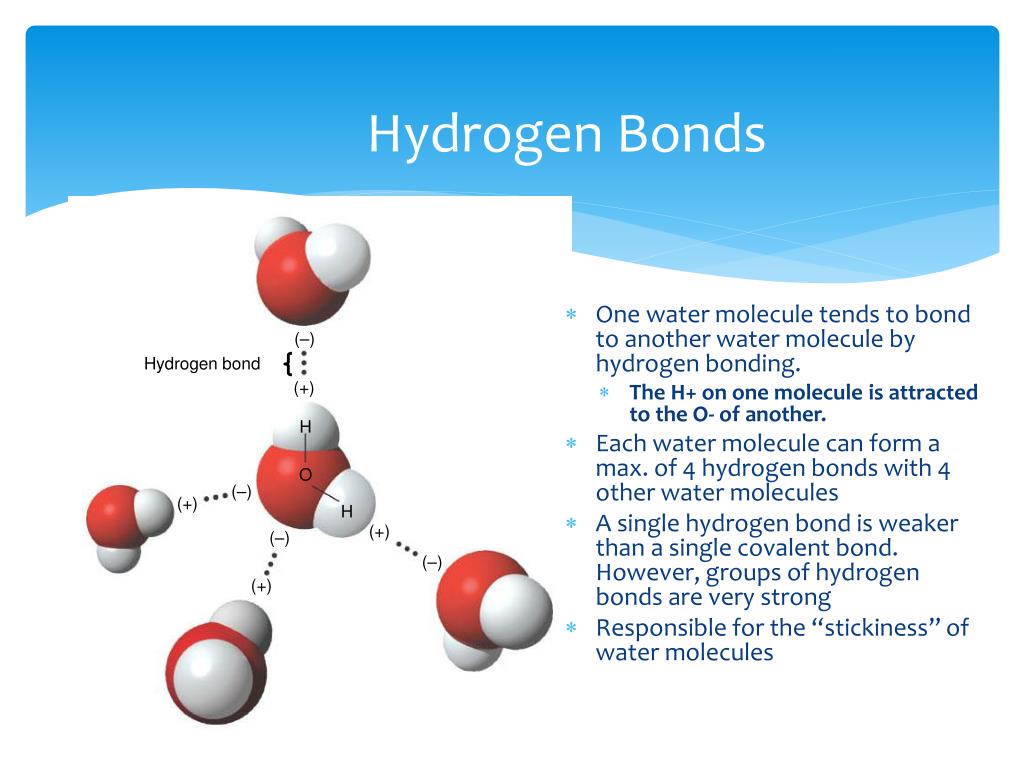
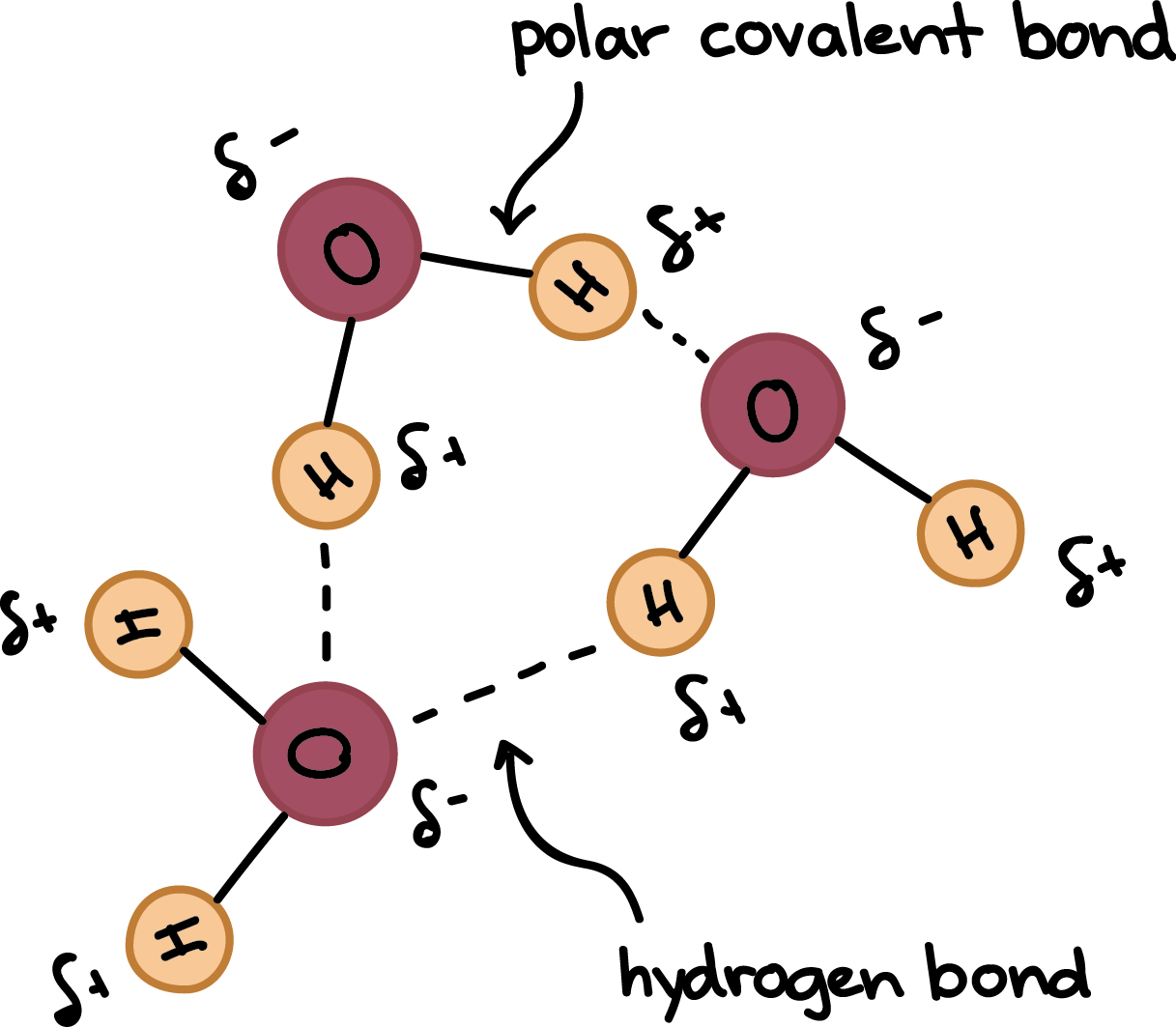Why Do Hydrogen Bonds Form Between Water Molecules - In water there are two lone pairs and two hydrogen atoms, allowing the formation of two hydrogen bonds. When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. Hydrogen bonding is an intermolecular force and. There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts. In ammonia, although there are three. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules.
When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. In water there are two lone pairs and two hydrogen atoms, allowing the formation of two hydrogen bonds. In ammonia, although there are three. There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. Hydrogen bonding is an intermolecular force and. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts.
Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts. There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. In ammonia, although there are three. When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. Hydrogen bonding is an intermolecular force and. In water there are two lone pairs and two hydrogen atoms, allowing the formation of two hydrogen bonds.
Hydrogen Bonds Between Water Molecules Diagram Science Onlin
There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. In ammonia, although there are three. In water there are two lone pairs and two hydrogen atoms, allowing the formation of two hydrogen bonds. Hydrogen bonding is an intermolecular force and. This allows for hydrogen bonding to occur.
SOLVED The diagram below shows hydrogen bonds between water molecules
There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. In ammonia, although there are three. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the.
Diagram of hydrogen bonding between two water molecules Download
Hydrogen bonding is an intermolecular force and. In water there are two lone pairs and two hydrogen atoms, allowing the formation of two hydrogen bonds. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen.
Water Review
In ammonia, although there are three. Hydrogen bonding is an intermolecular force and. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts. When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. In water.
Chapter 22 PROPERTIES OF WATER. ppt download
There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. Hydrogen bonding is an intermolecular force and. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and.
The Diagram Shows Hydrogen Bonds Between Water Molecules. Wh
In ammonia, although there are three. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts. When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. Hydrogen bonding is an intermolecular force and. In water.
The diagram shows hydrogen bonds between water molecules. Label the
When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. In water there are two lone pairs and two hydrogen atoms, allowing the formation of two hydrogen bonds. In ammonia, although there are three. Hydrogen bonding occurs in water due to the large electronegativity difference between.
Water Molecule Polarity And Hydrogen Bond Formation
Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts. When water freezes into ice, the h 2 o molecules form hydrogen bonds between each other and arrange themselves into an open lattice structure. Hydrogen bonding is an intermolecular force and. In water there are two lone pairs and.
Standard d. Explain the impact of water on life processes (i.e
In ammonia, although there are three. There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the.
Hydrogen Bonds Between Water Molecules Diagram Science Onlin
In ammonia, although there are three. There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. Hydrogen bonding is an intermolecular force and. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. In water there are two lone pairs and two hydrogen.
In Water There Are Two Lone Pairs And Two Hydrogen Atoms, Allowing The Formation Of Two Hydrogen Bonds.
There is a weak intermolecular force of attraction between the patially positively charged hydrogen atom of one molecule and the partially negativly. This allows for hydrogen bonding to occur between oxygens and hydrogens of different water molecules. Hydrogen bonding is an intermolecular force and. In ammonia, although there are three.
When Water Freezes Into Ice, The H 2 O Molecules Form Hydrogen Bonds Between Each Other And Arrange Themselves Into An Open Lattice Structure.
Hydrogen bonding occurs in water due to the large electronegativity difference between the hydrogen atoms and the oxygen atom.the oxygen attracts.