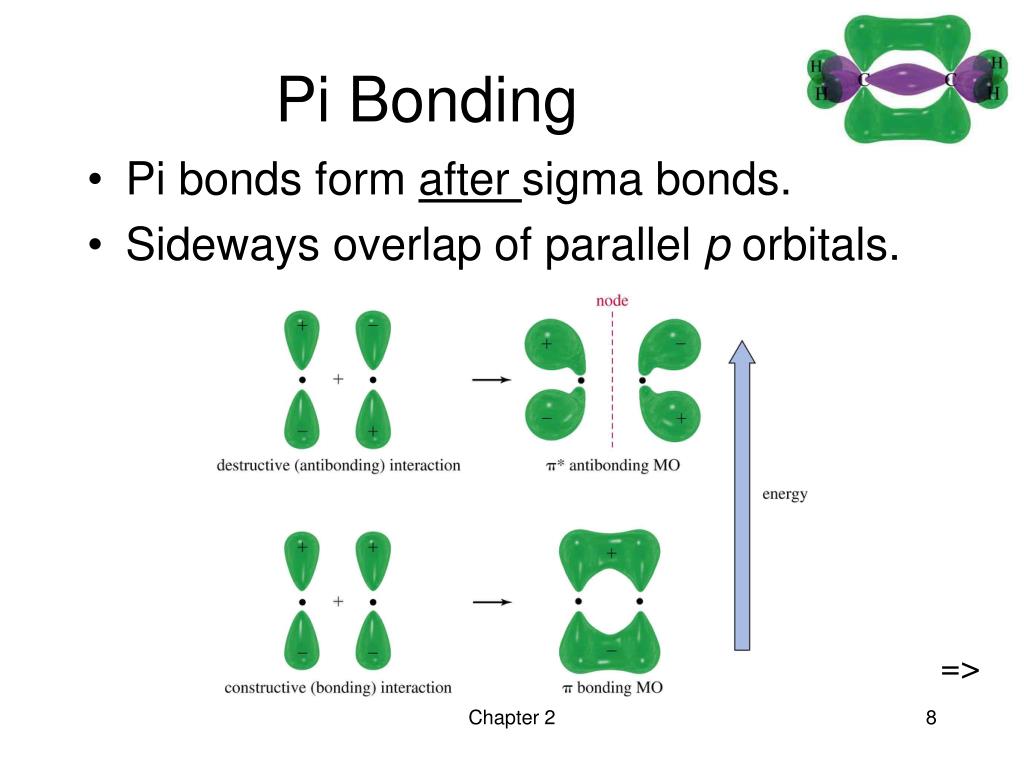
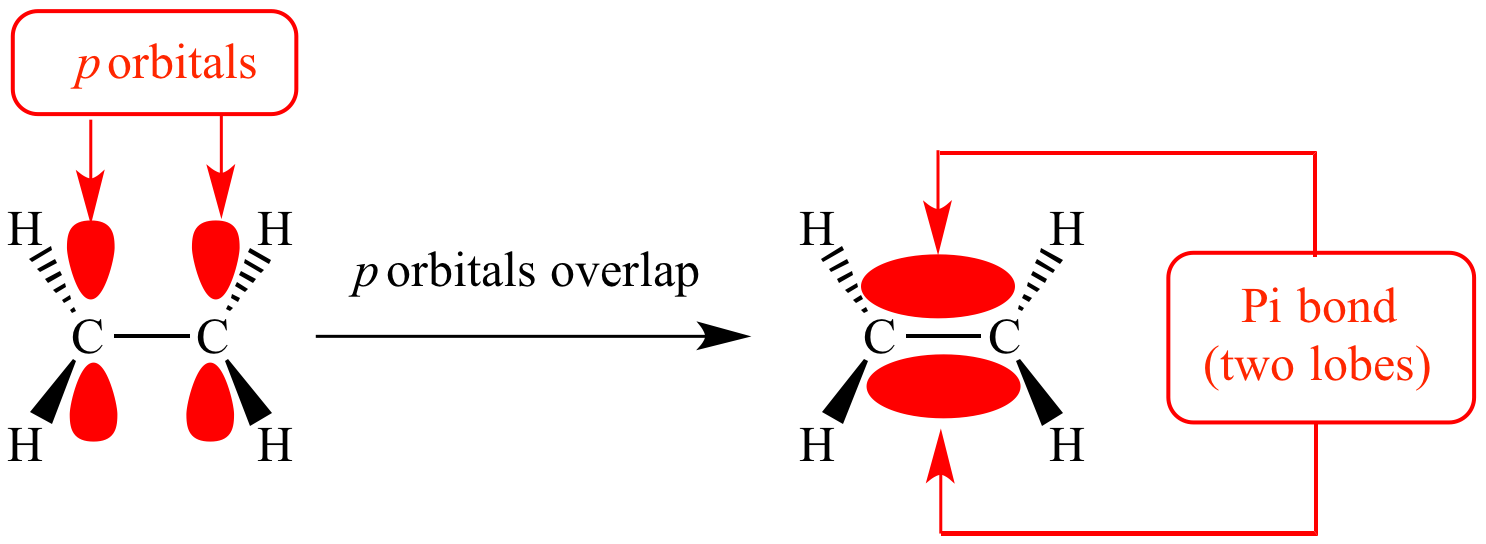
Which Orbitals Form A Pi Bond - The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed at the beginning. Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of. Recommended videos shapes of orbitals what is an orbital? What are the 4 atomic orbitals? There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. What is molecular orbital theory? The orbitals in an atom are organized into different layers or electron shells.
What is molecular orbital theory? The orbitals in an atom are organized into different layers or electron shells. The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of. What are the 4 atomic orbitals? Recommended videos shapes of orbitals what is an orbital? Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed at the beginning. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f.
The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed at the beginning. What is molecular orbital theory? What are the 4 atomic orbitals? Recommended videos shapes of orbitals what is an orbital? There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. The orbitals in an atom are organized into different layers or electron shells. Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of.
PPT Chemical Bonding and Molecular Structure (Ch. 10) PowerPoint
The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed at the beginning. Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the.
Sigma and pi bonds
What is molecular orbital theory? What are the 4 atomic orbitals? The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. Recommended videos shapes of orbitals what.
Sigma (δ) and Pi ((π) Bond (ALevel) ChemistryStudent
Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. Recommended videos shapes of orbitals what is an orbital? The orbitals in an atom are organized into.
Bonding And Antibonding Pi Orbitals Master Organic Chemistry
The orbitals in an atom are organized into different layers or electron shells. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. What is molecular orbital theory? What are the 4 atomic orbitals? The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed.
Hybridization. ppt download
Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. The difference between orbit and orbitals is important to understand for any budding chemistry student or even.
Bonding Theory Overview ppt download
The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed at the beginning. The orbitals in an atom are organized into different layers or electron shells. Recommended videos shapes of orbitals what is an orbital? Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of.
PPT Chapter 2 Structure and Properties of Organic Molecules
The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of. Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. The molecular orbital theory (often abbreviated to mot) is a theory on chemical bonding developed at.
Illustrated Glossary of Organic Chemistry Pi bond
What is molecular orbital theory? Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. The orbitals in an atom are organized into different layers or electron.
Sigma and pi bonds. Overlapping of Atomic orbitals. Vector illustration
What is molecular orbital theory? There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. Recommended videos shapes of orbitals what is an orbital? What are the 4 atomic orbitals? Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair.
Chapters 9 and 11 study guide
There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f. Recommended videos shapes of orbitals what is an orbital? The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of. The molecular orbital theory (often abbreviated to mot).
Recommended Videos Shapes Of Orbitals What Is An Orbital?
Orbitals in physics and chemistry is a mathematical function depicting the wave nature of an electron or a pair of electrons present. What is molecular orbital theory? What are the 4 atomic orbitals? The orbitals in an atom are organized into different layers or electron shells.
The Molecular Orbital Theory (Often Abbreviated To Mot) Is A Theory On Chemical Bonding Developed At The Beginning.
The difference between orbit and orbitals is important to understand for any budding chemistry student or even professionals in the field of. There are four types of orbitals that you should know (sharp, principal, diffuse and fundamental) with s, p, d and f.
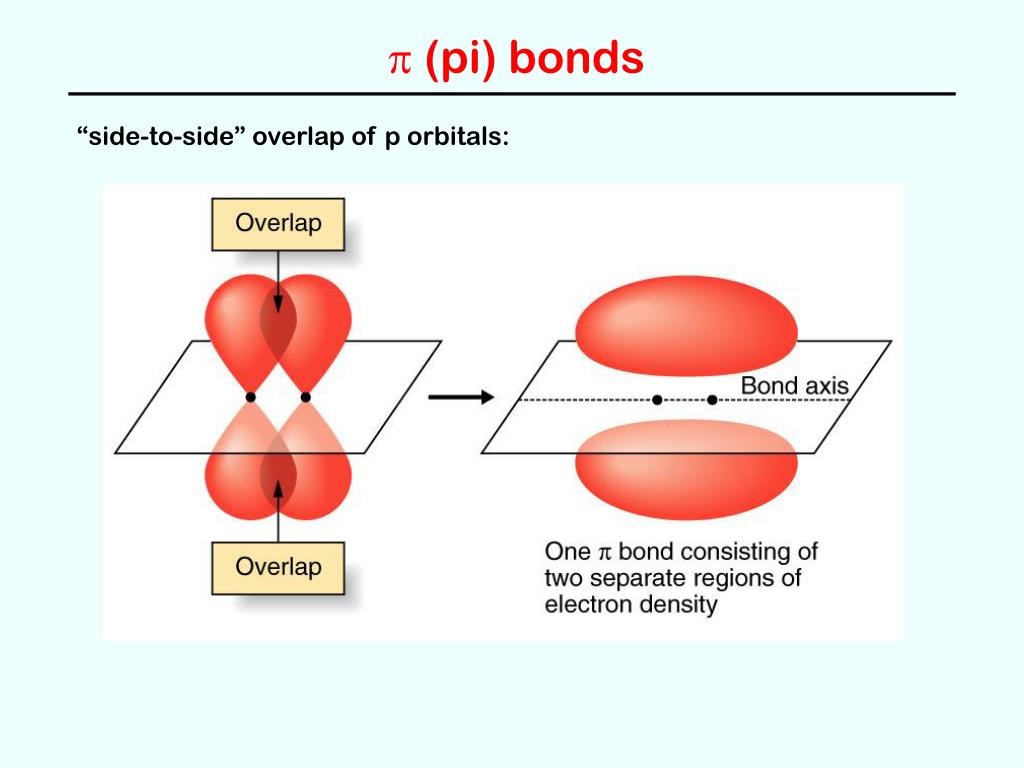
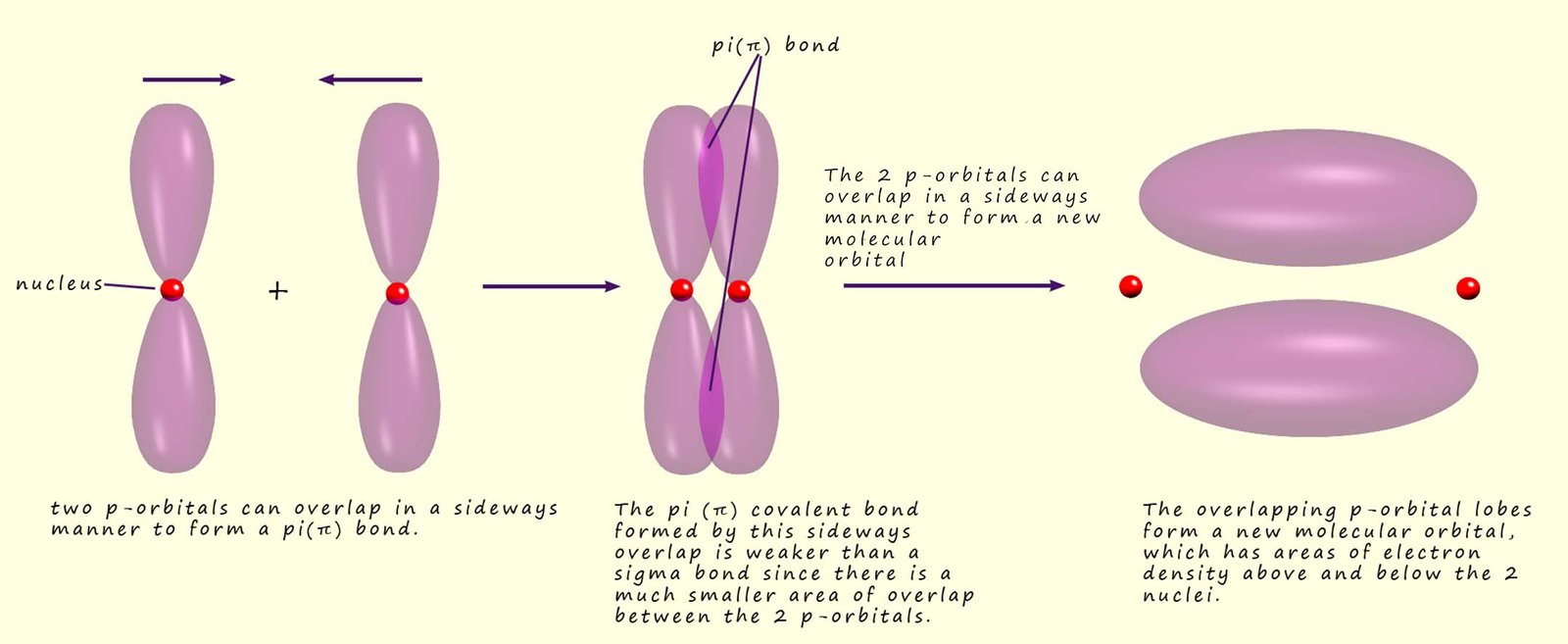
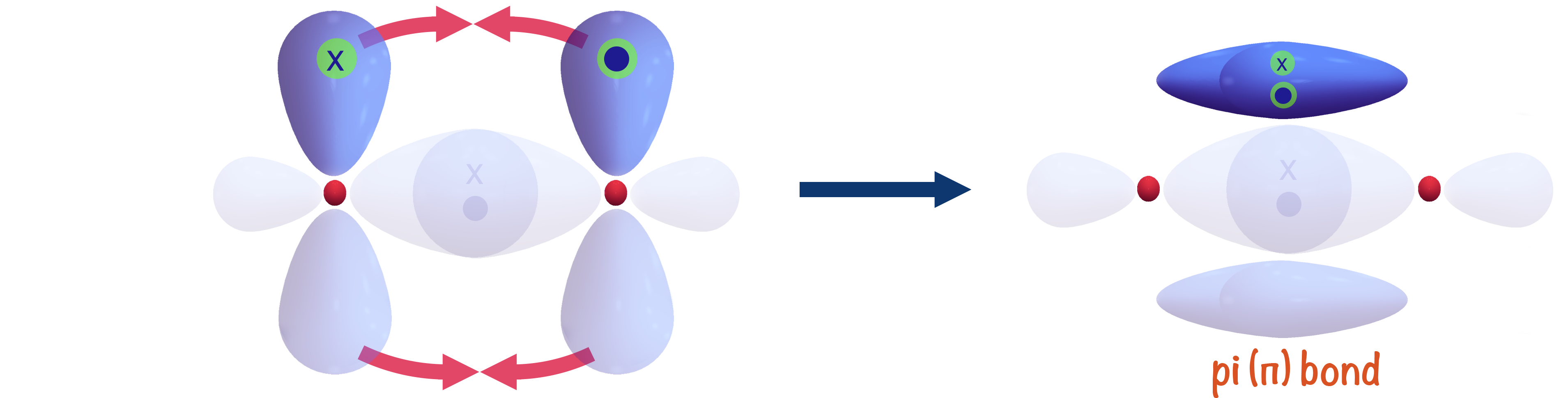
+A+covalent+bond+formed+by+the+parallel+(side+by+side)+overlap+of+two+p+orbitals..jpg)
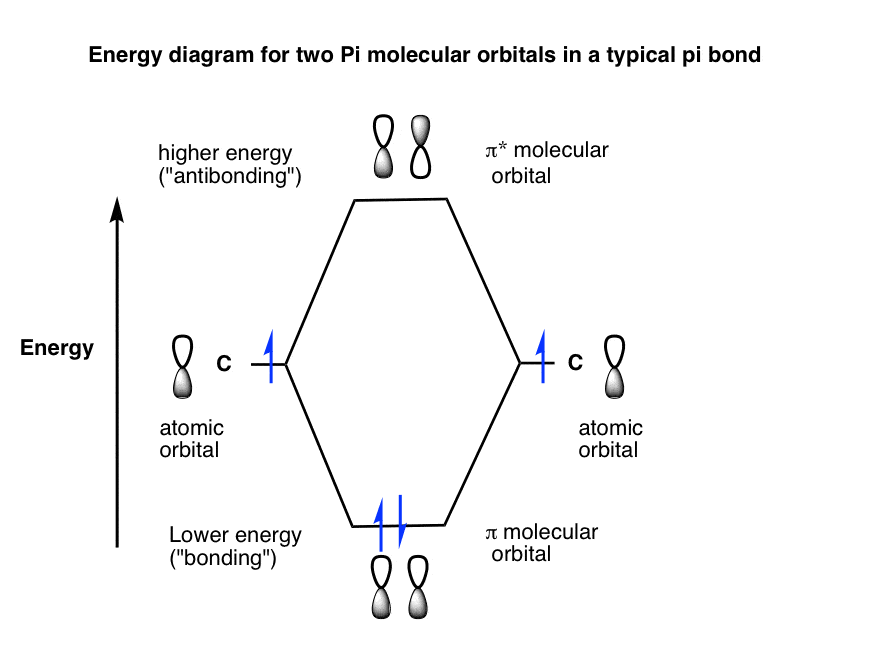
+bonds%2C+SIDE-BY-SIDE+overlap..jpg)