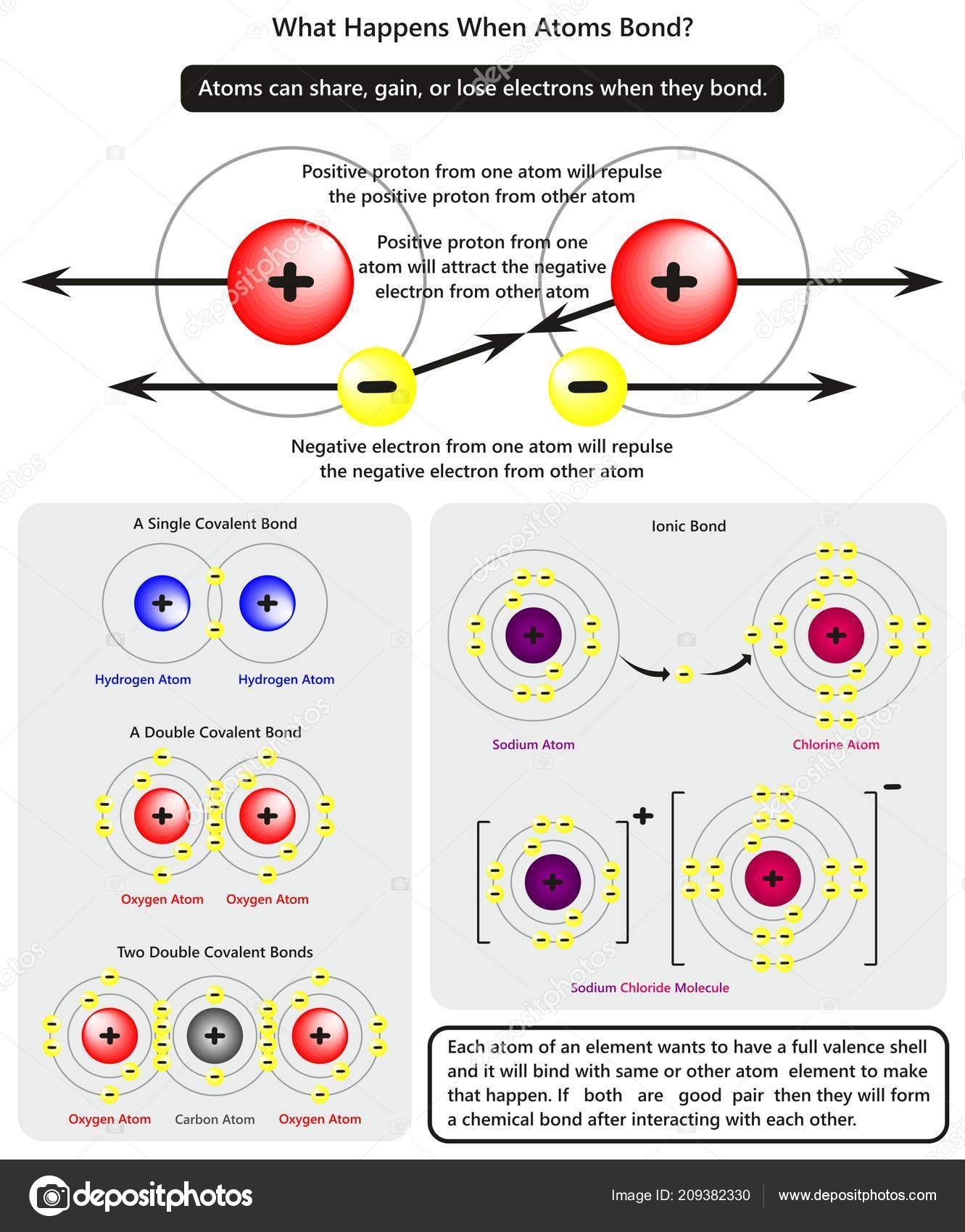
What Happens When Two Atoms Form A Chemical Bond - Yes, an atom with 6 protons and 6 neutrons can form. The force of attraction between electron of one atom. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. Bromine is a diatomic molecule, which means the two bromine. An atom that has gained an electron has a negative charge and is called an anion. A covalent bond is formed when two bromine atoms react with each other. All chemistry is based on sharing electrons. During formation of bond,the atoms come close,then their are two possibilities 1.
Yes, an atom with 6 protons and 6 neutrons can form. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. During formation of bond,the atoms come close,then their are two possibilities 1. A covalent bond is formed when two bromine atoms react with each other. The force of attraction between electron of one atom. An atom that has gained an electron has a negative charge and is called an anion. Bromine is a diatomic molecule, which means the two bromine. All chemistry is based on sharing electrons.
During formation of bond,the atoms come close,then their are two possibilities 1. Bromine is a diatomic molecule, which means the two bromine. An atom that has gained an electron has a negative charge and is called an anion. A covalent bond is formed when two bromine atoms react with each other. The force of attraction between electron of one atom. All chemistry is based on sharing electrons. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. Yes, an atom with 6 protons and 6 neutrons can form.
What Happens Atoms Bond Infographic Diagram Showing How Electrons
A covalent bond is formed when two bromine atoms react with each other. Bromine is a diatomic molecule, which means the two bromine. The force of attraction between electron of one atom. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. An atom that has gained an electron has a negative charge.
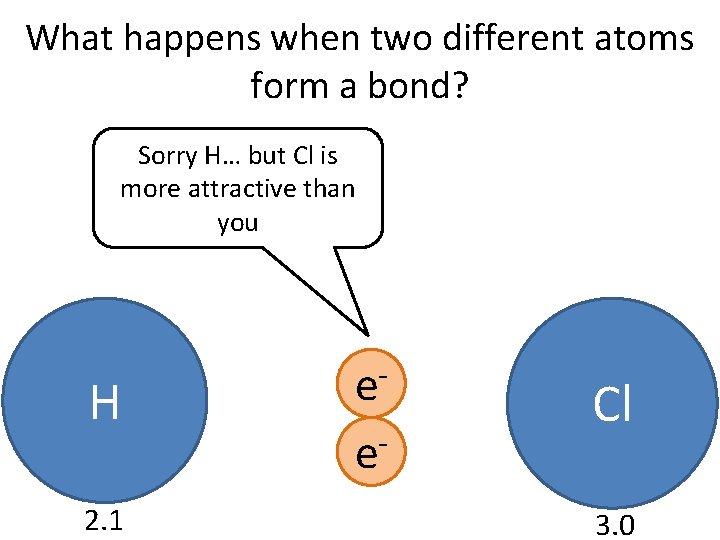
Do Now Why do atoms form chemical bonds, and what happens during the
The force of attraction between electron of one atom. Bromine is a diatomic molecule, which means the two bromine. An atom that has gained an electron has a negative charge and is called an anion. A covalent bond is formed when two bromine atoms react with each other. A chemical bond forms when two atoms transfer or share outer electrons.
INTRAMOLECULAR FORCES CHEMICAL BONDS Chemical bond force that
All chemistry is based on sharing electrons. Yes, an atom with 6 protons and 6 neutrons can form. The force of attraction between electron of one atom. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. An atom that has gained an electron has a negative charge and is called an anion.
CHEMICAL BONDING & FORMULAS ppt download
Bromine is a diatomic molecule, which means the two bromine. During formation of bond,the atoms come close,then their are two possibilities 1. An atom that has gained an electron has a negative charge and is called an anion. Yes, an atom with 6 protons and 6 neutrons can form. A covalent bond is formed when two bromine atoms react with.
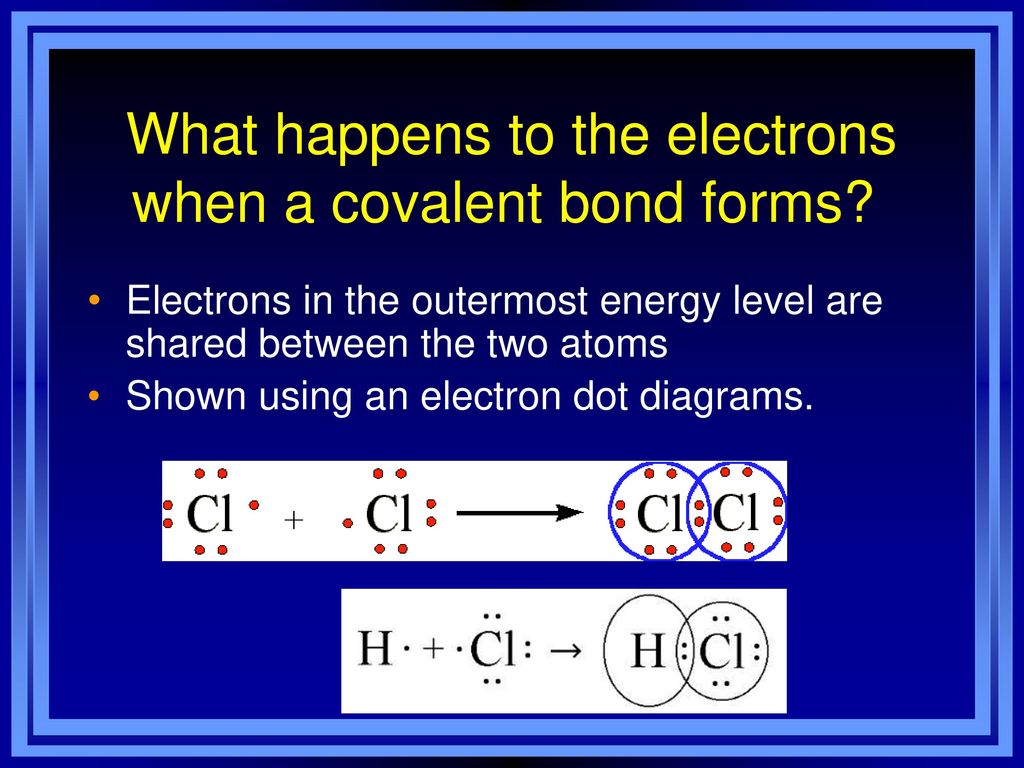
Aim What are covalent bonds and how are they formed? ppt download
An atom that has gained an electron has a negative charge and is called an anion. Yes, an atom with 6 protons and 6 neutrons can form. During formation of bond,the atoms come close,then their are two possibilities 1. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. Bromine is a diatomic.
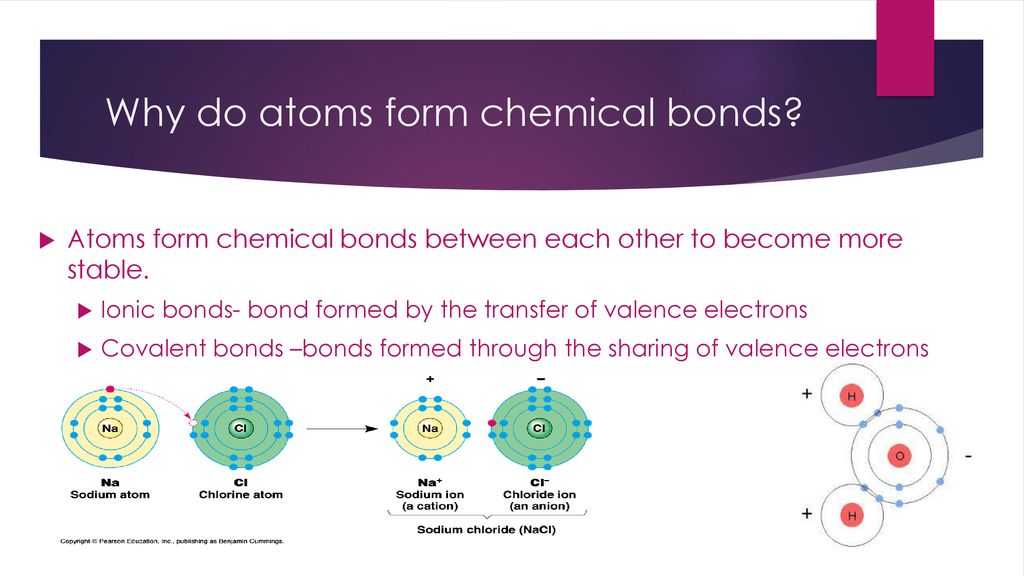
Introduction to Chemical Bonding ppt download
The force of attraction between electron of one atom. All chemistry is based on sharing electrons. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. An atom that has gained an electron has a negative charge and is called an anion. A covalent bond is formed when two bromine atoms react with.
CHEMICAL BONDING What happens when Bonds are Formed
During formation of bond,the atoms come close,then their are two possibilities 1. A covalent bond is formed when two bromine atoms react with each other. Bromine is a diatomic molecule, which means the two bromine. All chemistry is based on sharing electrons. Yes, an atom with 6 protons and 6 neutrons can form.
Chemical Bonds and Compounds ppt download
Bromine is a diatomic molecule, which means the two bromine. Yes, an atom with 6 protons and 6 neutrons can form. A covalent bond is formed when two bromine atoms react with each other. The force of attraction between electron of one atom. During formation of bond,the atoms come close,then their are two possibilities 1.
Do Now Why do atoms form chemical bonds, and what happens during the
A covalent bond is formed when two bromine atoms react with each other. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. An atom that has gained an electron has a negative charge and is called an anion. Bromine is a diatomic molecule, which means the two bromine. All chemistry is based.
Chemistry of Life Chapter ppt download
During formation of bond,the atoms come close,then their are two possibilities 1. An atom that has gained an electron has a negative charge and is called an anion. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. Bromine is a diatomic molecule, which means the two bromine. All chemistry is based on.
Yes, An Atom With 6 Protons And 6 Neutrons Can Form.
A covalent bond is formed when two bromine atoms react with each other. An atom that has gained an electron has a negative charge and is called an anion. A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. All chemistry is based on sharing electrons.
During Formation Of Bond,The Atoms Come Close,Then Their Are Two Possibilities 1.
Bromine is a diatomic molecule, which means the two bromine. The force of attraction between electron of one atom.




..jpg)
