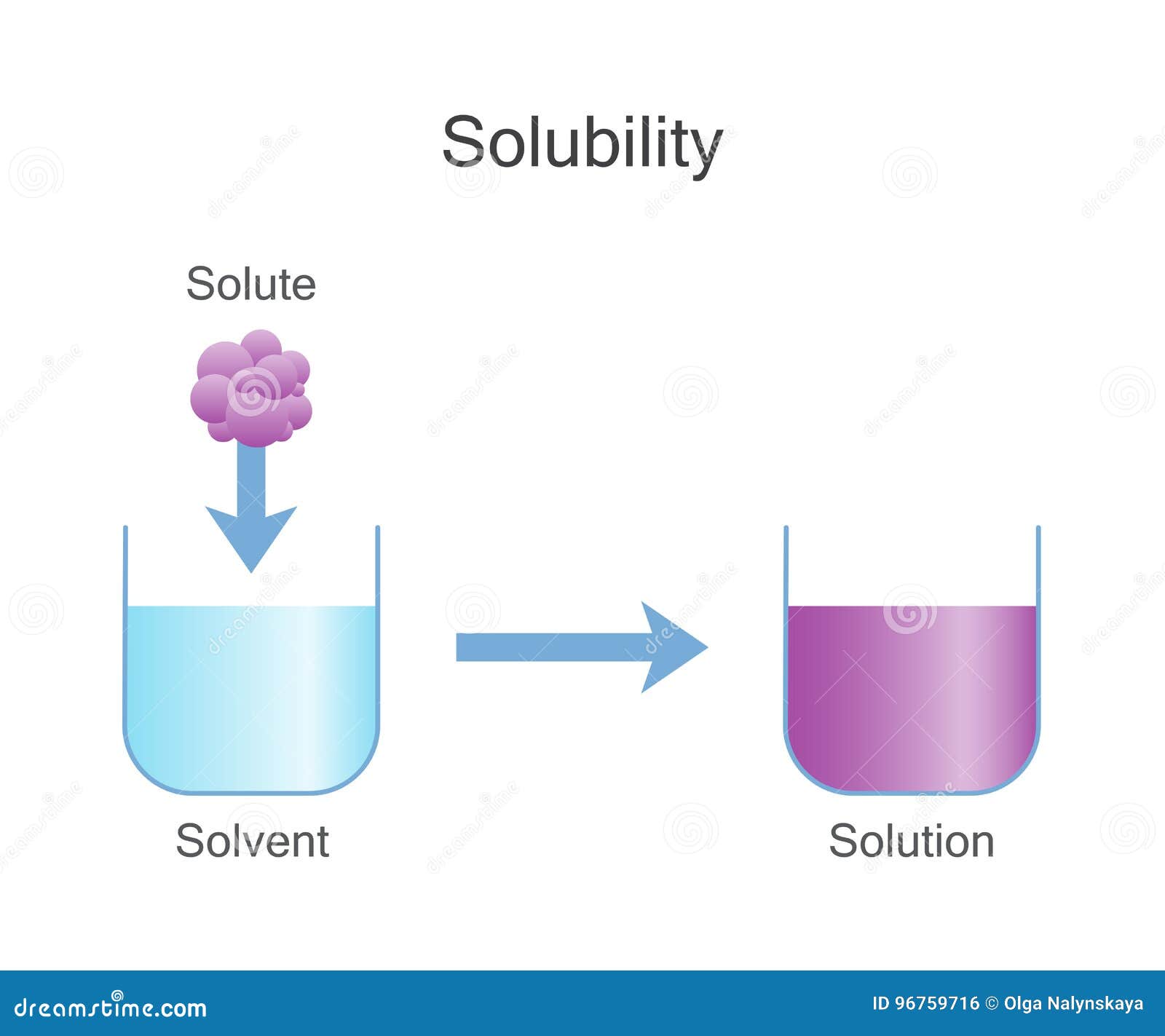
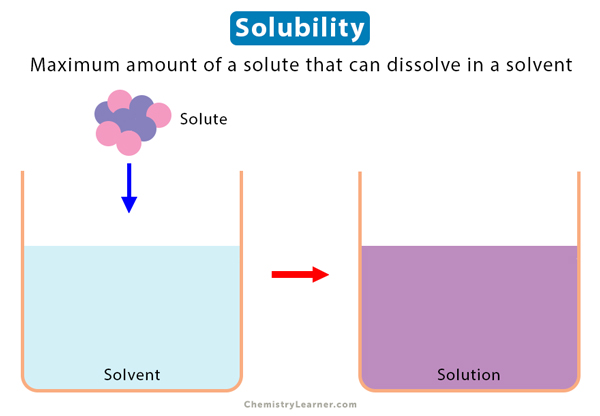
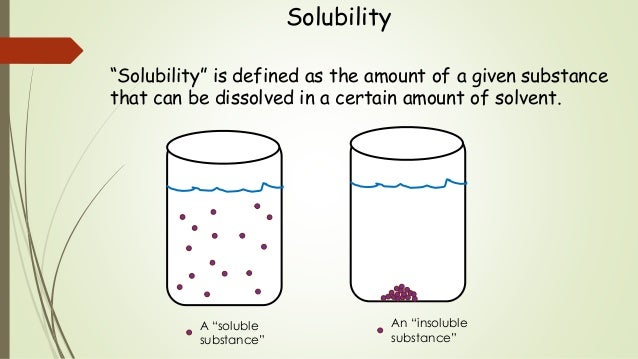
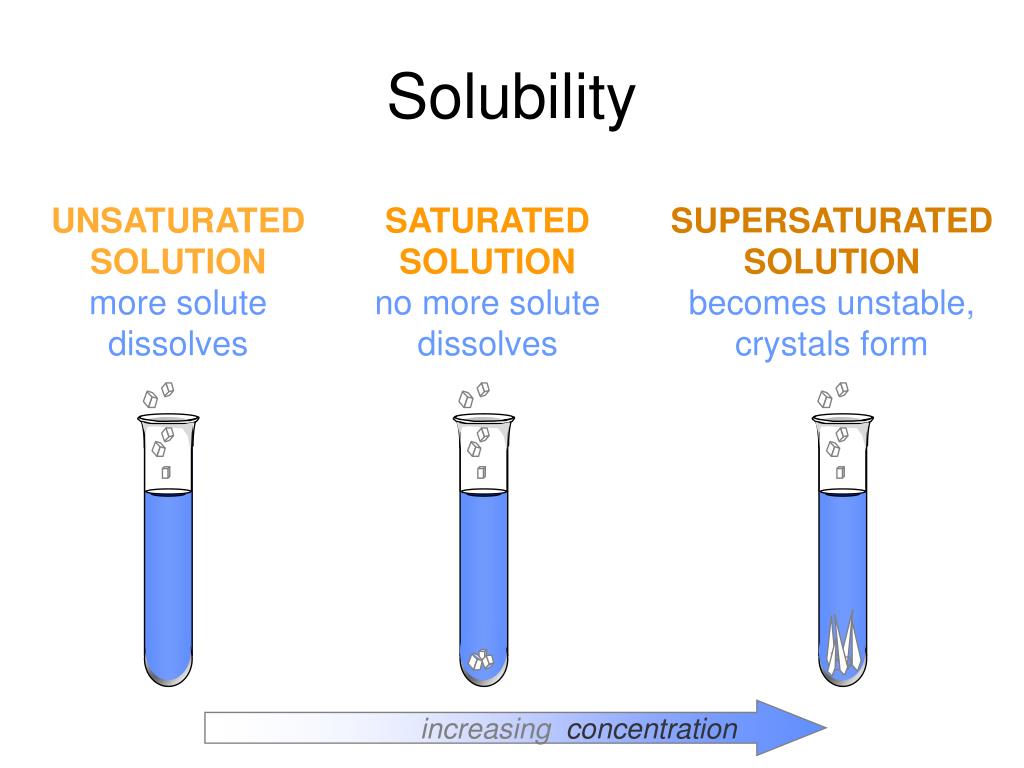
Solubility Sheet - Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). The process through which a. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. In such an equilibrium, le chatelier's. Insolubility is the opposite property,. This is the property that allows things like sugar molecules to dissolve in. Solubility is the ability of a solute to dissolve in a solvent to form a solution.
Insolubility is the opposite property,. The process through which a. Solubility is the ability of a solute to dissolve in a solvent to form a solution. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. This is the property that allows things like sugar molecules to dissolve in. In such an equilibrium, le chatelier's. Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature.
Solubility is the ability of a solute to dissolve in a solvent to form a solution. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). The process through which a. Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. In such an equilibrium, le chatelier's. This is the property that allows things like sugar molecules to dissolve in. Insolubility is the opposite property,.
Solid Solubility In Phase Diagrams Solid Solution Phase Diag
This is the property that allows things like sugar molecules to dissolve in. Insolubility is the opposite property,. Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature. The process through which a. Solubility is the ability of a solute to dissolve in a solvent to form a.
What is Solubility Definition of Solubility
Solubility is the ability of a solute to dissolve in a solvent to form a solution. Insolubility is the opposite property,. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). Solubility is defined as the upper limit of solute that can be dissolved in a.
Solubility Vector Illustration. Labeled Solute, Solvent and Solution
Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). In such an equilibrium, le chatelier's. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. This is the property that allows things like sugar molecules.
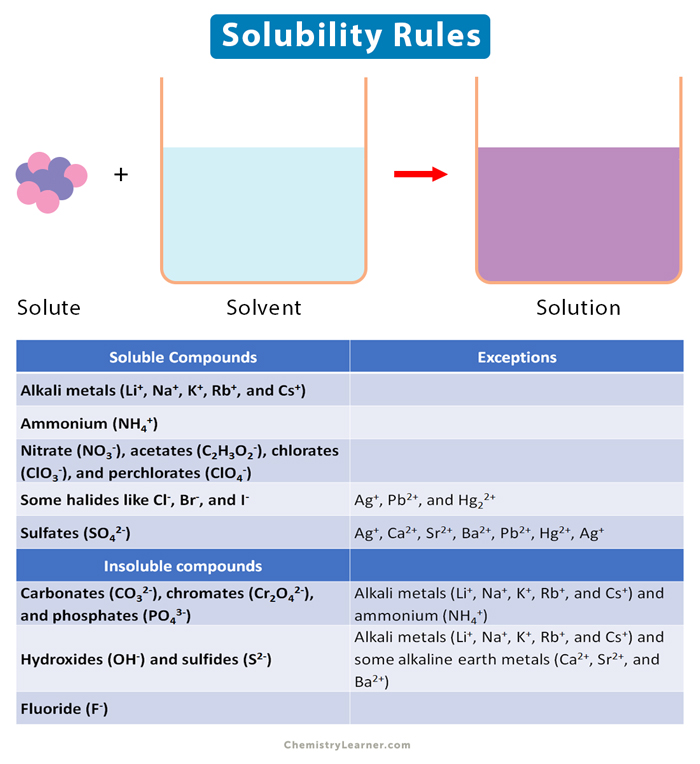
Solubility Rules Definition, Examples, and Table
Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). In chemistry, solubility is the ability of a substance, the solute, to form a solution.
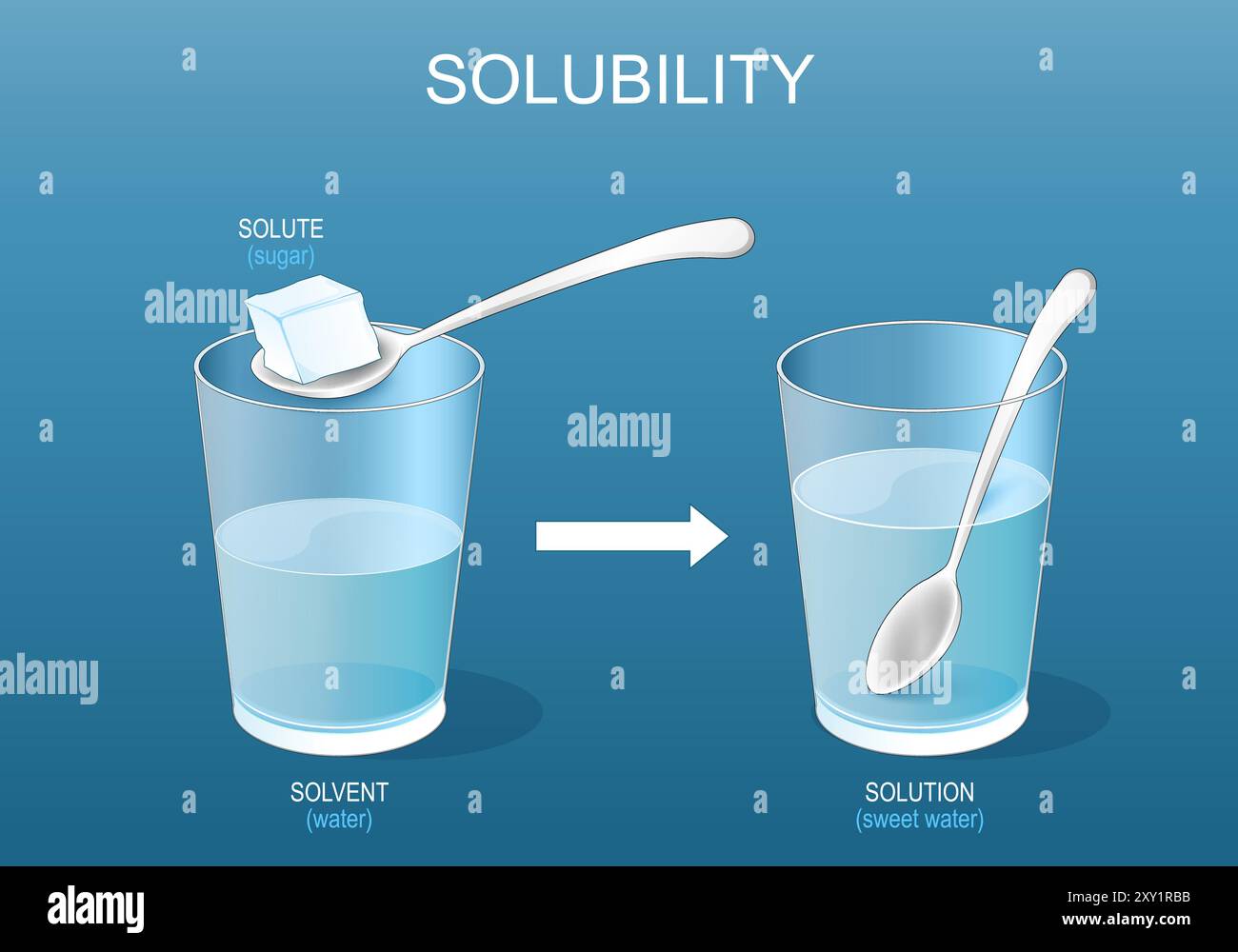
Dissolution sugar Stock Vector Images Alamy
Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. Insolubility is the opposite property,. The process through which a. In chemistry, solubility is the ability of.
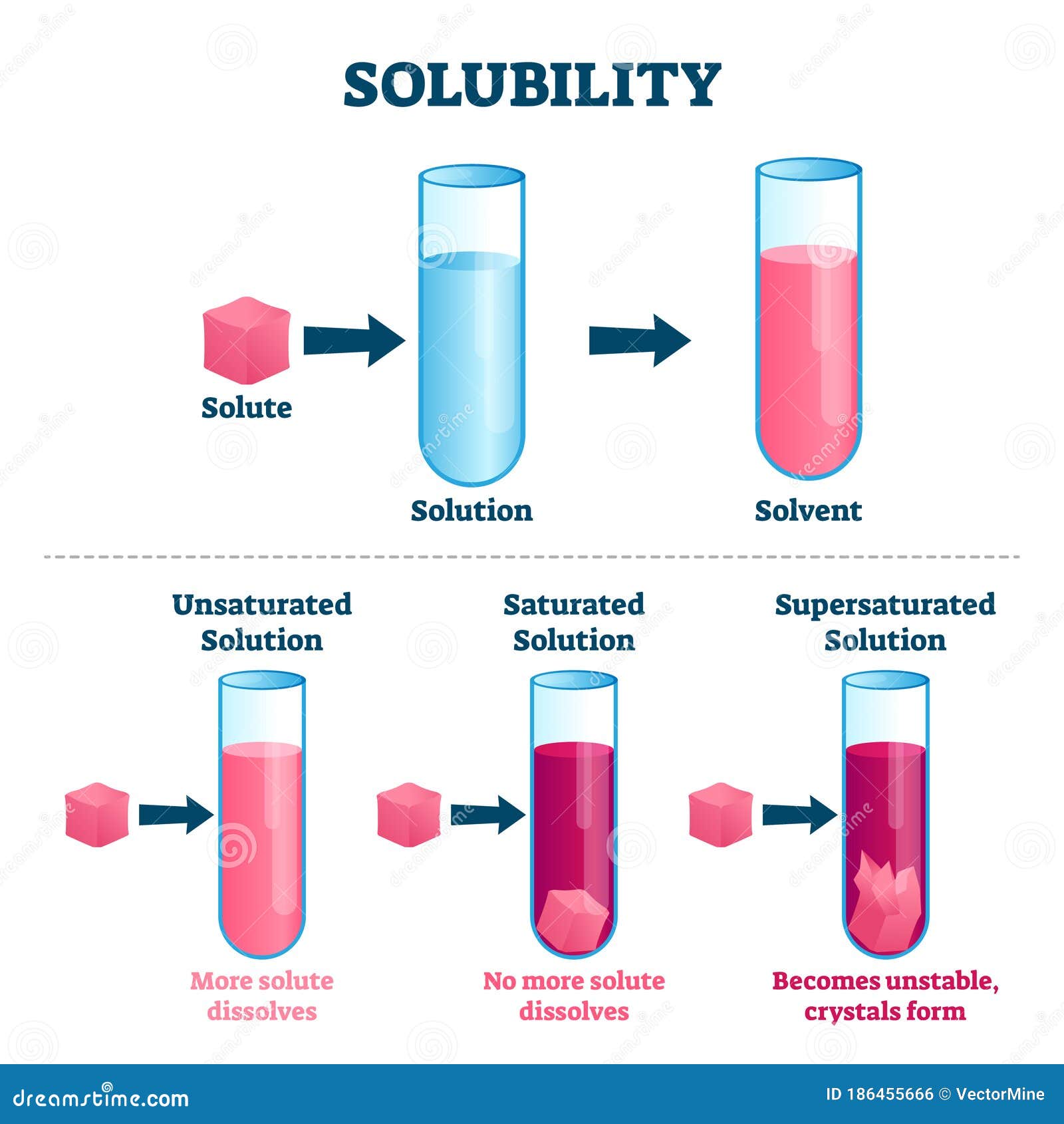
Solubility General Chemistry Lecture Slides Docsity
Insolubility is the opposite property,. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. The process through which a. This is the property that allows things like sugar.
Example Of Soluble
Insolubility is the opposite property,. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. This is the property that allows things like sugar molecules to dissolve in. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Solubility.
Solubility
The process through which a. Insolubility is the opposite property,. This is the property that allows things like sugar molecules to dissolve in. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Solubility is the ability of a solute to dissolve in a solvent to form a solution.
PPT Solubility PowerPoint Presentation, free download ID1115118
Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. The process through which a. Solubility is the ability of a solute to dissolve in a solvent to form.
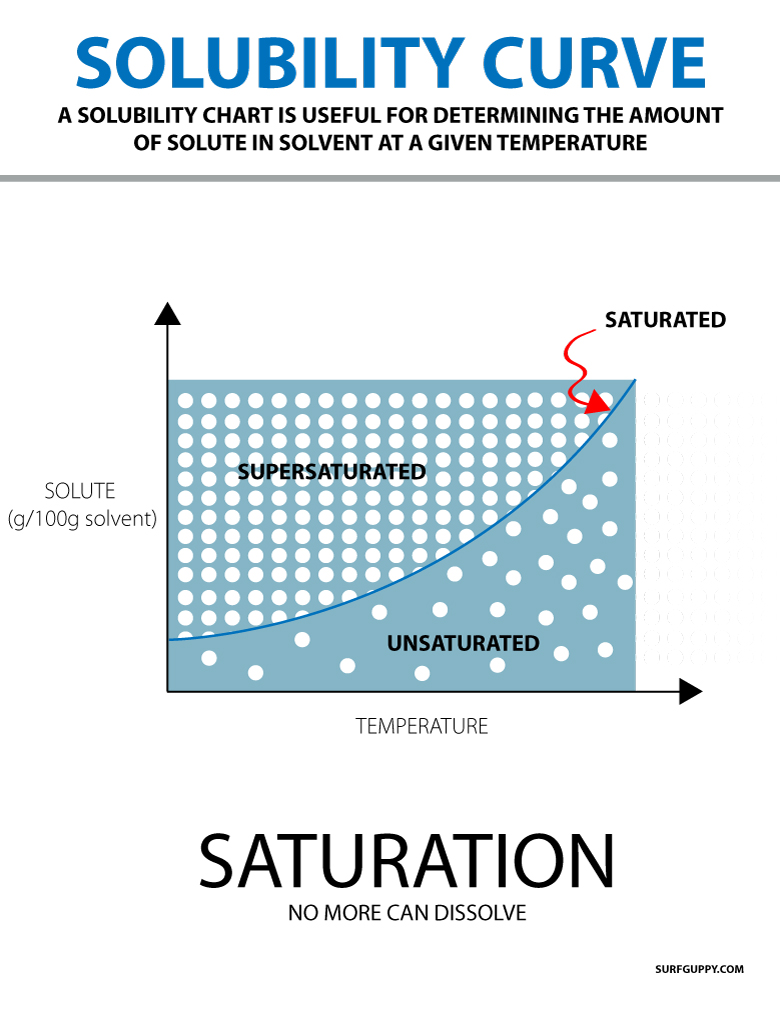
Solubility Surfguppy Chemistry made easy visual learning
Solubility is the ability of a solute to dissolve in a solvent to form a solution. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a.
Solubility Is The Ability Of A Solute To Dissolve In A Solvent To Form A Solution.
Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. This is the property that allows things like sugar molecules to dissolve in. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent).
In Chemistry, Solubility Is The Ability Of A Substance, The Solute, To Form A Solution With Another Substance, The Solvent.
The process through which a. Insolubility is the opposite property,. In such an equilibrium, le chatelier's.