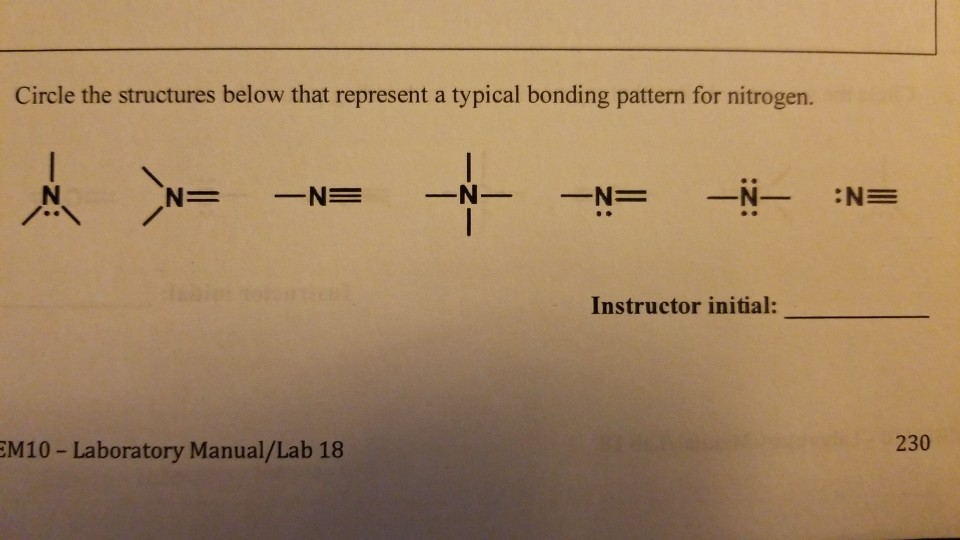
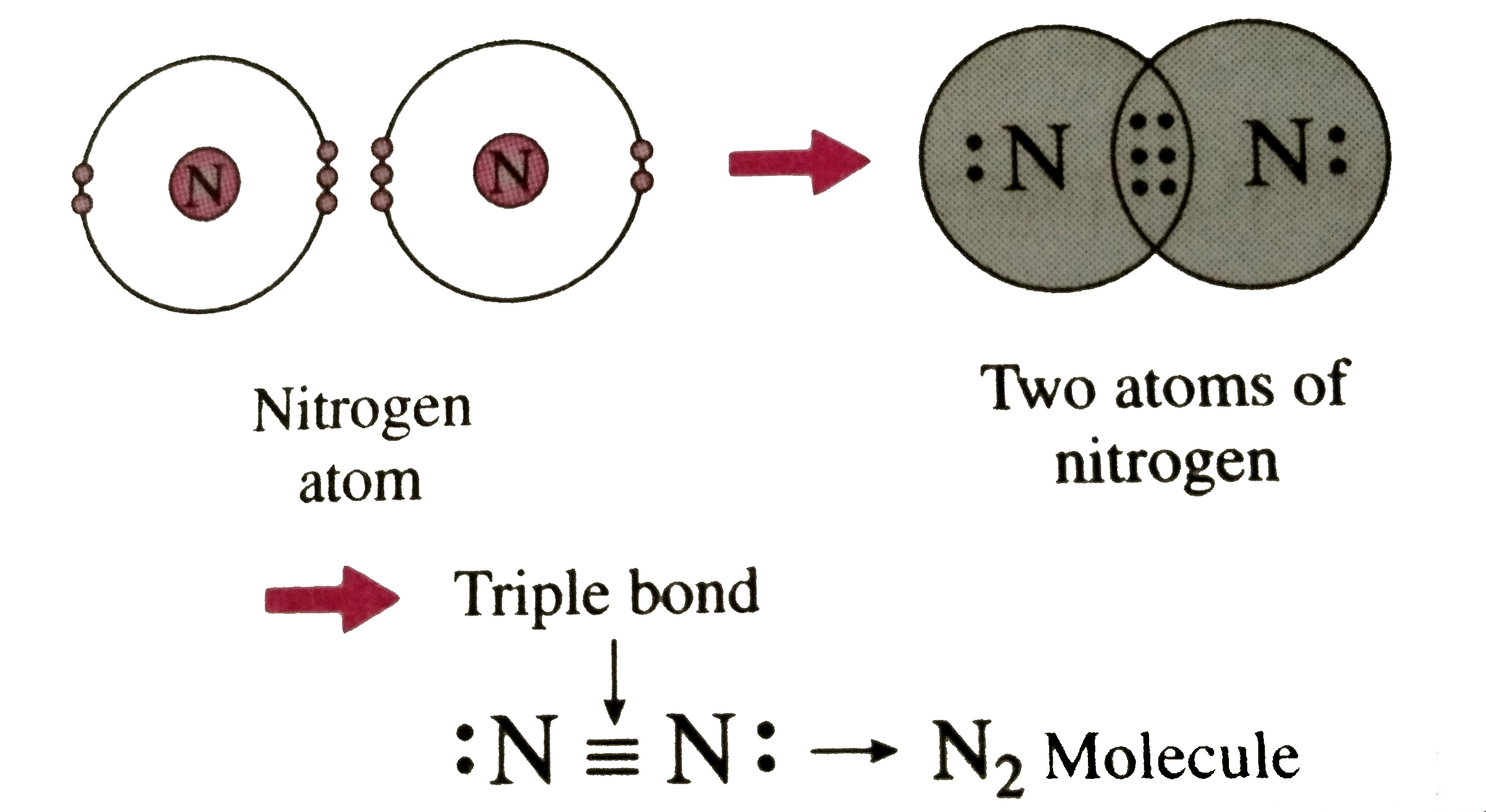How Many Bonds Does Nitrogen Form - Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. There is a total of three covalent. However, with a positive net charge,. Ammonia is a nitrogen atom bonded to three hydrogen atoms. Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. How many covalent bonds are in ammonia? That means it needs 3 bonds. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration.
However, with a positive net charge,. How many covalent bonds are in ammonia? As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. There is a total of three covalent. That means it needs 3 bonds. Ammonia is a nitrogen atom bonded to three hydrogen atoms. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only.
Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. However, with a positive net charge,. That means it needs 3 bonds. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. Ammonia is a nitrogen atom bonded to three hydrogen atoms. There is a total of three covalent. Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. How many covalent bonds are in ammonia?
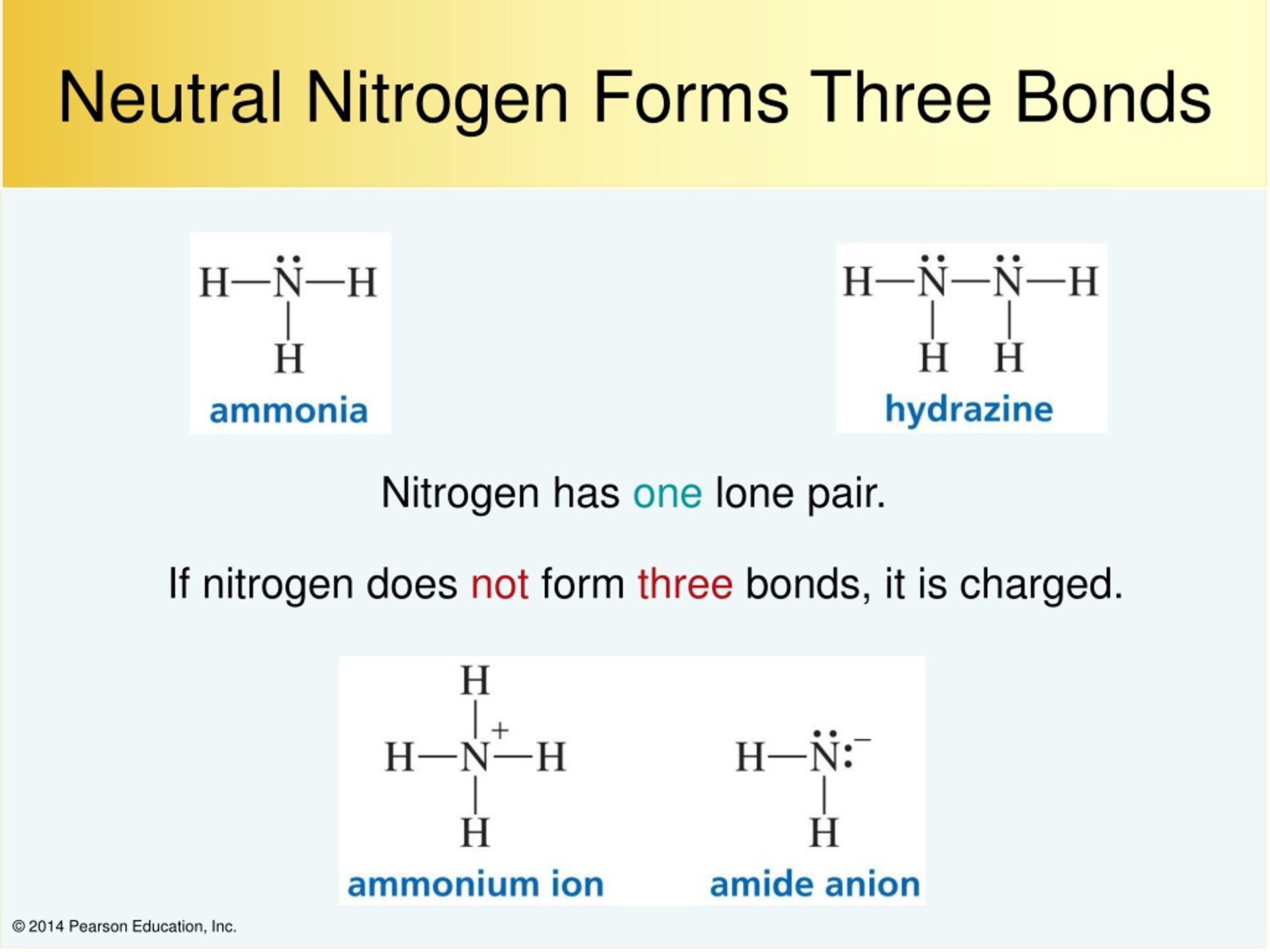
Nitrogen Covalent Bond
However, with a positive net charge,. Ammonia is a nitrogen atom bonded to three hydrogen atoms. How many covalent bonds are in ammonia? That means it needs 3 bonds. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons.
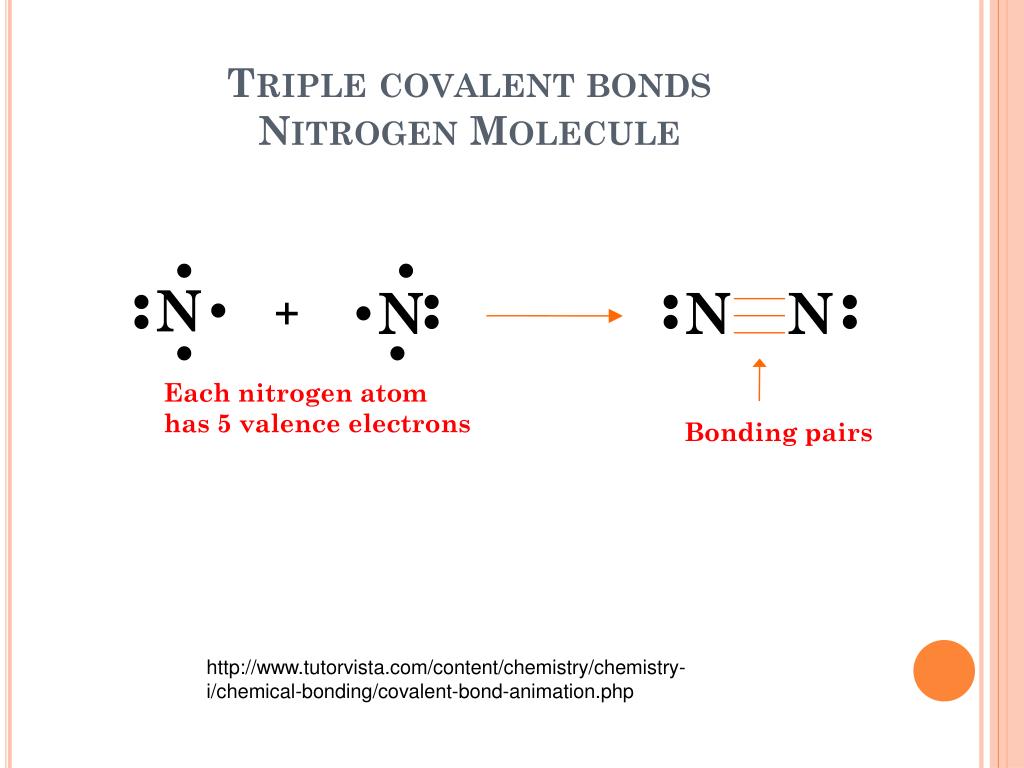
Covalent Bond N2
That means it needs 3 bonds. Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. There is a total of three covalent. An atom of nitrogen typically forms 3 covalent.
PPT Remembering General Chemistry Electronic Structure and Bonding
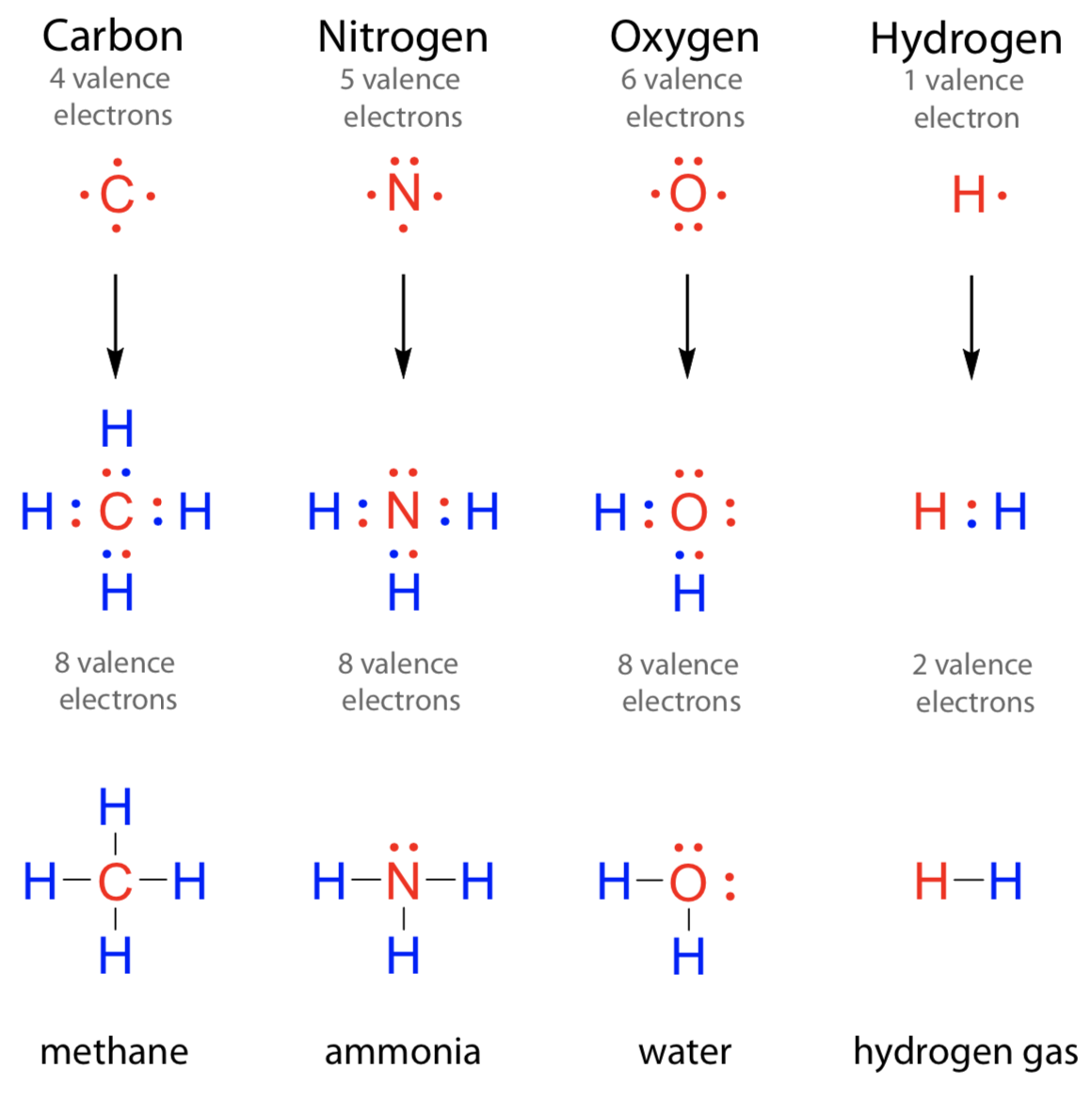
Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. Ammonia is a nitrogen atom bonded to three hydrogen atoms. How many covalent bonds are in ammonia? There is a total of three covalent. Nitrogen can't hybridise to form 5 bonds, since it literally has only one s.
Describe the formation of nitrogen molecule.
There is a total of three covalent. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. However, with a positive net charge,. Ammonia is a nitrogen atom bonded to three hydrogen atoms. That means it needs 3 bonds.
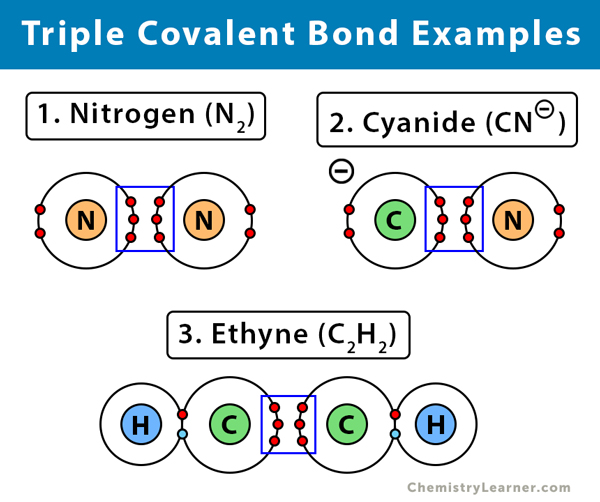
Nitrogen Molecule Covalent Bond
Ammonia is a nitrogen atom bonded to three hydrogen atoms. Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. However, with a positive net charge,. Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are.
Covalent Bond N2
Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. Ammonia is a nitrogen atom bonded to three hydrogen atoms. However, with a positive net.
LabXchange
Ammonia is a nitrogen atom bonded to three hydrogen atoms. Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. How many covalent bonds are in ammonia? As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. However, with a.
PPT Covalent Bonding and Chemical Bonds PowerPoint Presentation, free
There is a total of three covalent. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. However, with a positive net charge,. Ammonia is a nitrogen atom bonded to three hydrogen atoms. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration.
Solved How many bonds does nitrogen typically form? Does
Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. Ammonia is a nitrogen atom bonded to three hydrogen atoms. There is a total of three covalent. Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state.
PPT COVALENT BONDING PowerPoint Presentation, free download ID5128236
As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. That means it needs 3 bonds. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. However, with a positive net charge,. Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine,.
How Many Covalent Bonds Are In Ammonia?
Hydrogen bonds occur when hydrogen is bonded to an electronegative atom like oxygen, nitrogen, or fluorine, and in c3h9n, there are only. There is a total of three covalent. That means it needs 3 bonds. Ammonia is a nitrogen atom bonded to three hydrogen atoms.
An Atom Of Nitrogen Typically Forms 3 Covalent Bonds To Achieve A Stable Electron Configuration.
Nitrogen can't hybridise to form 5 bonds, since it literally has only one s and three p's in its ground state energy level. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. However, with a positive net charge,.