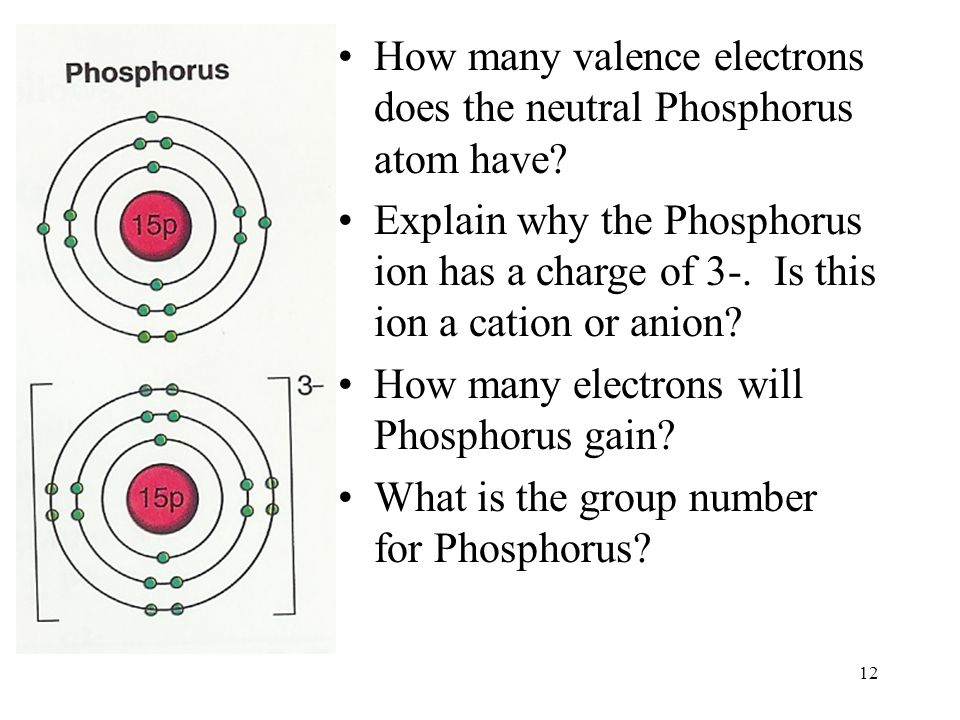
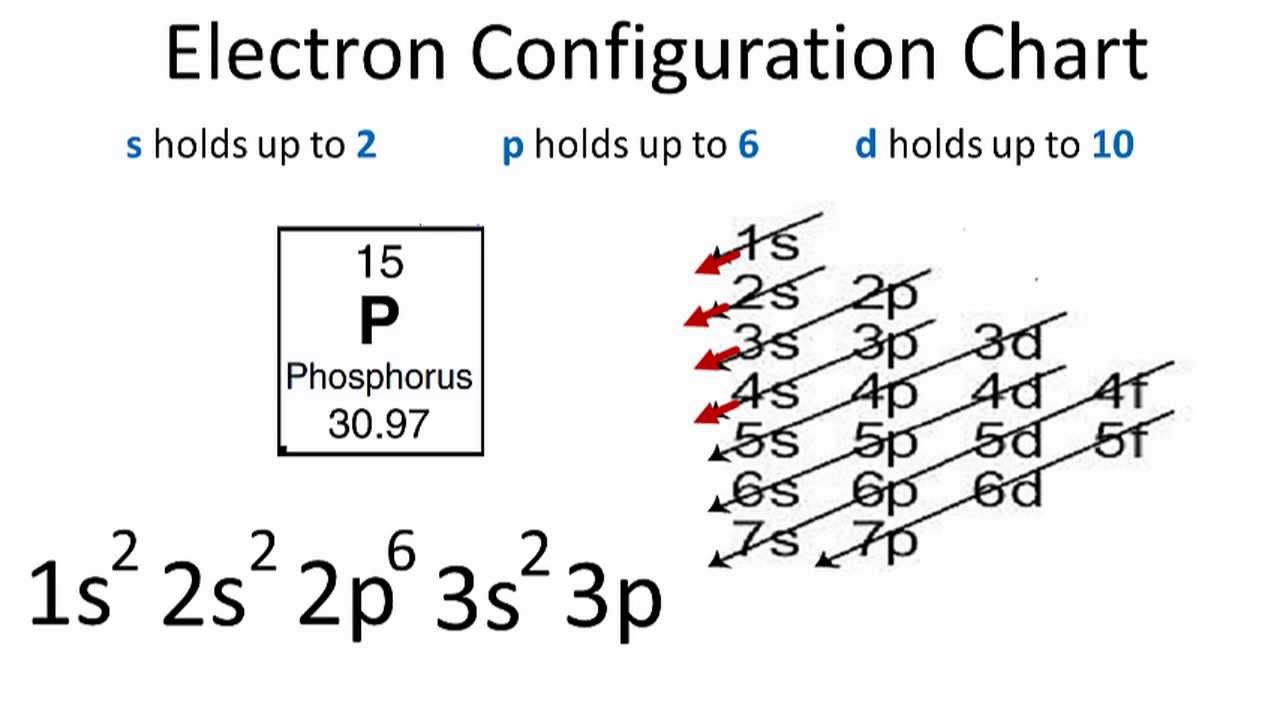
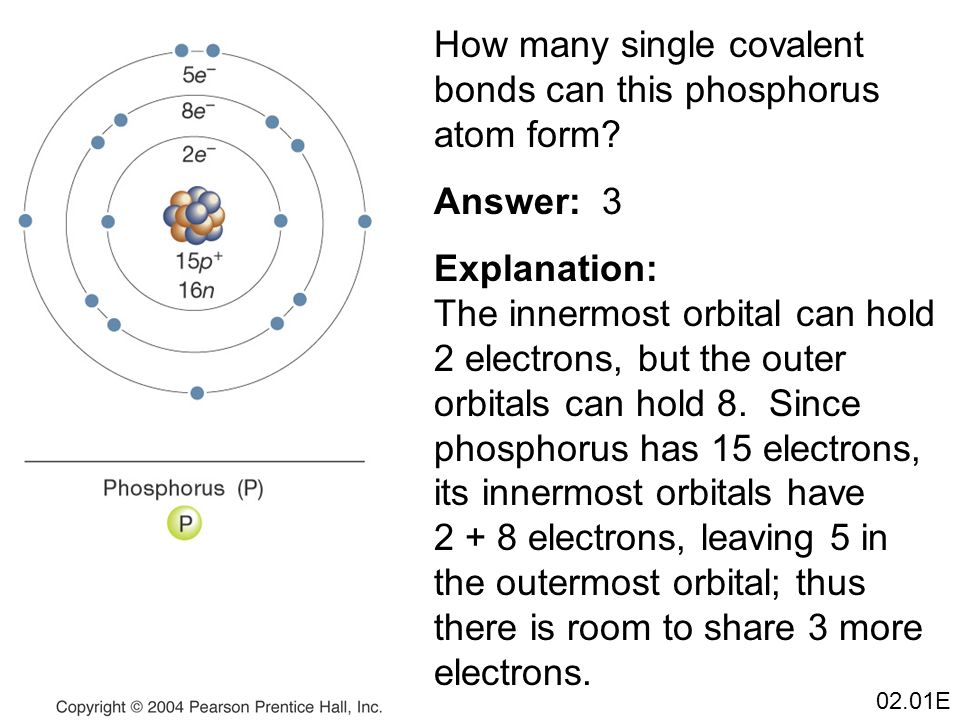
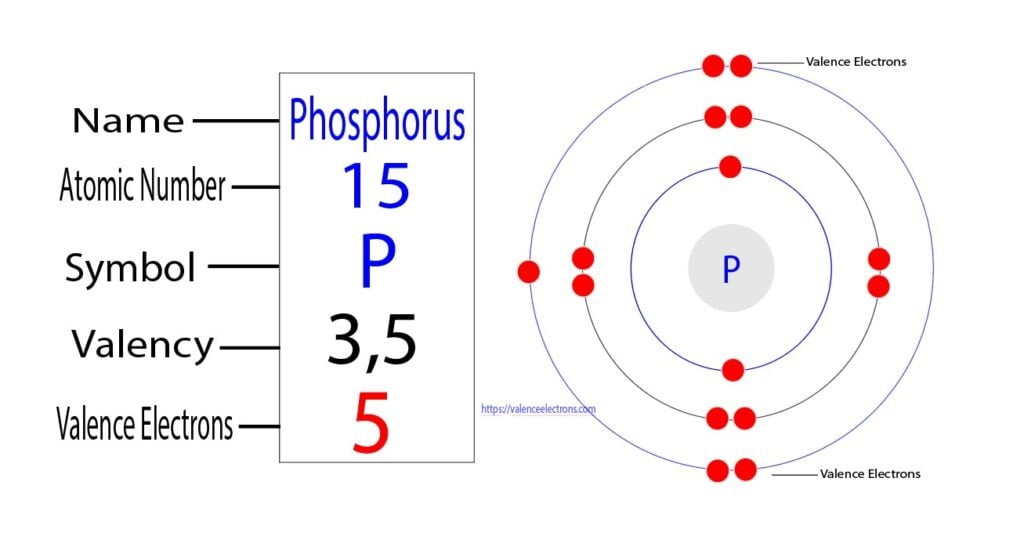
How Many Bonds Can Phosphorus Form - How phosphorus forms 2 types of bonds with oxygen i.e. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus can form 5 bonds like in the case of phosphate. While nitrogen is known to make a maximum of 4 bonds (3. Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that.
Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. While nitrogen is known to make a maximum of 4 bonds (3. How phosphorus forms 2 types of bonds with oxygen i.e. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Phosphorus can form 5 bonds like in the case of phosphate.
Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. While nitrogen is known to make a maximum of 4 bonds (3. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus can form 5 bonds like in the case of phosphate. Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. How phosphorus forms 2 types of bonds with oxygen i.e.
Init 1/16/2017 by Daniel R. Barnes ppt download
Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Phosphorus can form 5 bonds like in the case of phosphate. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Calling.
Symbol of Phosphorus Archives Dynamic Periodic Table of Elements and
Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. While nitrogen is known to make a.
Phosphorus Atom
How phosphorus forms 2 types of bonds with oxygen i.e. While nitrogen is known to make a maximum of 4 bonds (3. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. Phosphorus only 'needs' three more.
LabXchange
5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: How phosphorus forms 2 types of bonds with oxygen i.e. While nitrogen.
Phosphorus Electric Dot Diagram
5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. Phosphorus can form 5 bonds like in.
Phosphorus Atom Model Project
How phosphorus forms 2 types of bonds with oxygen i.e. Phosphorus can form 5 bonds like in the case of phosphate. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. While nitrogen is known to make a maximum of 4 bonds (3. Calling electrostatic attractions ionic bonds creates too many false parallels to.
[Solved] PCl3F2 How many single bonds does phosphorus have it? Course
I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Phosphorus can form 5 bonds like in the case of phosphate. While.
How Many Valence Electrons Does PH3 (Phosphine) Have?
Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. While nitrogen is known to make a maximum of 4 bonds (3. 5 in $\ce {p4o10}$ and 3 in $\ce {p4o6}$ when only three are needed to complete. Phosphorus can form 5 bonds like in the case of phosphate. Phosphorus only.
Phosphorus Lewis Dot Diagram
While nitrogen is known to make a maximum of 4 bonds (3. Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. How phosphorus forms 2 types of bonds with oxygen i.e. 5.
What Are The Different Industrial Uses Of Phosphorus
Phosphorus can form 5 bonds like in the case of phosphate. How phosphorus forms 2 types of bonds with oxygen i.e. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that. 5 in $\ce {p4o10}$ and 3 in.
5 In $\Ce {P4O10}$ And 3 In $\Ce {P4O6}$ When Only Three Are Needed To Complete.
While nitrogen is known to make a maximum of 4 bonds (3. Calling electrostatic attractions ionic bonds creates too many false parallels to covalent bonds, which, in turn, generates barriers for. Phosphorus can form 5 bonds like in the case of phosphate. Phosphorus only 'needs' three more electrons to get a full valence shell of eight, but you'll notice that.
How Phosphorus Forms 2 Types Of Bonds With Oxygen I.e.
I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: