Fda Form 483 Definition - Learn more about the fda’s role in reviewing, approving,. Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to. Call the fda consumer complaint coordinator for your state or region.
Learn more about the fda’s role in reviewing, approving,. Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. Call the fda consumer complaint coordinator for your state or region. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to.
Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Call the fda consumer complaint coordinator for your state or region. Learn more about the fda’s role in reviewing, approving,.
PPT Handling Regulatory Inspections PowerPoint Presentation ID5770979
Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. Learn more about the fda’s role in reviewing, approving,. Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to. The fda is responsible for protecting the public.
FDA Form 483 Issued to Eli Lilly Regarding Zyprexa FDA 483s
The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. Learn more about the fda’s role in reviewing, approving,. Fda uses science and data to ensure that.
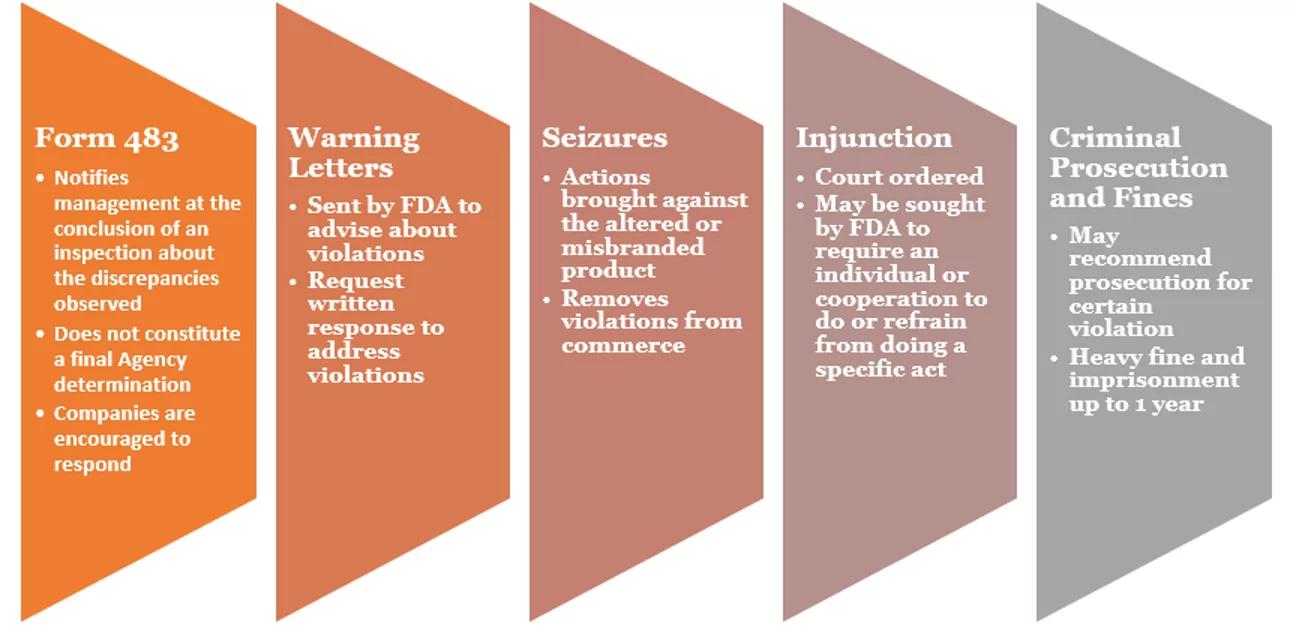
FDA Form 483 Observations and FDA Warning Letters What’s the Difference?
Learn more about the fda’s role in reviewing, approving,. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Call the fda consumer complaint coordinator for your state or region. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public.
Fda 483 Response Template
The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Call the fda consumer complaint coordinator for your state or region. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. Fda fulfills this responsibility by ensuring.
eBrief All You Need to Know about FDA Form 483 and Warning Letters
The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Call the fda consumer complaint coordinator for your state or region. Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to. Learn more about the fda’s.
FDA Form 483 A Guide to Assessing and Avoiding Violations
Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Call the fda consumer complaint coordinator for your state or region. Learn more about the fda’s.
LOGO
Learn more about the fda’s role in reviewing, approving,. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. Recalls, market withdrawals, & safety alerts the list below provides information.
483 vs Warning Letter What’s The Difference? EMMA International
Learn more about the fda’s role in reviewing, approving,. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about certain. Fda fulfills this responsibility by ensuring the security.
FDA Form 483 Warning Letters How to Handle, Form, Example » Pharmaguddu
The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. Recalls, market withdrawals, & safety alerts the list below provides information gathered from press releases and other public notices about.
US FDA Form 483 to Aurobindo Pharma Ltd Unit VI
Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to.
Learn More About The Fda’s Role In Reviewing, Approving,.
The fda is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological. Call the fda consumer complaint coordinator for your state or region. Fda uses science and data to ensure that approved drugs are of a high quality, safe, and effective. Fda fulfills this responsibility by ensuring the security of the food supply and by fostering development of medical products to respond to.