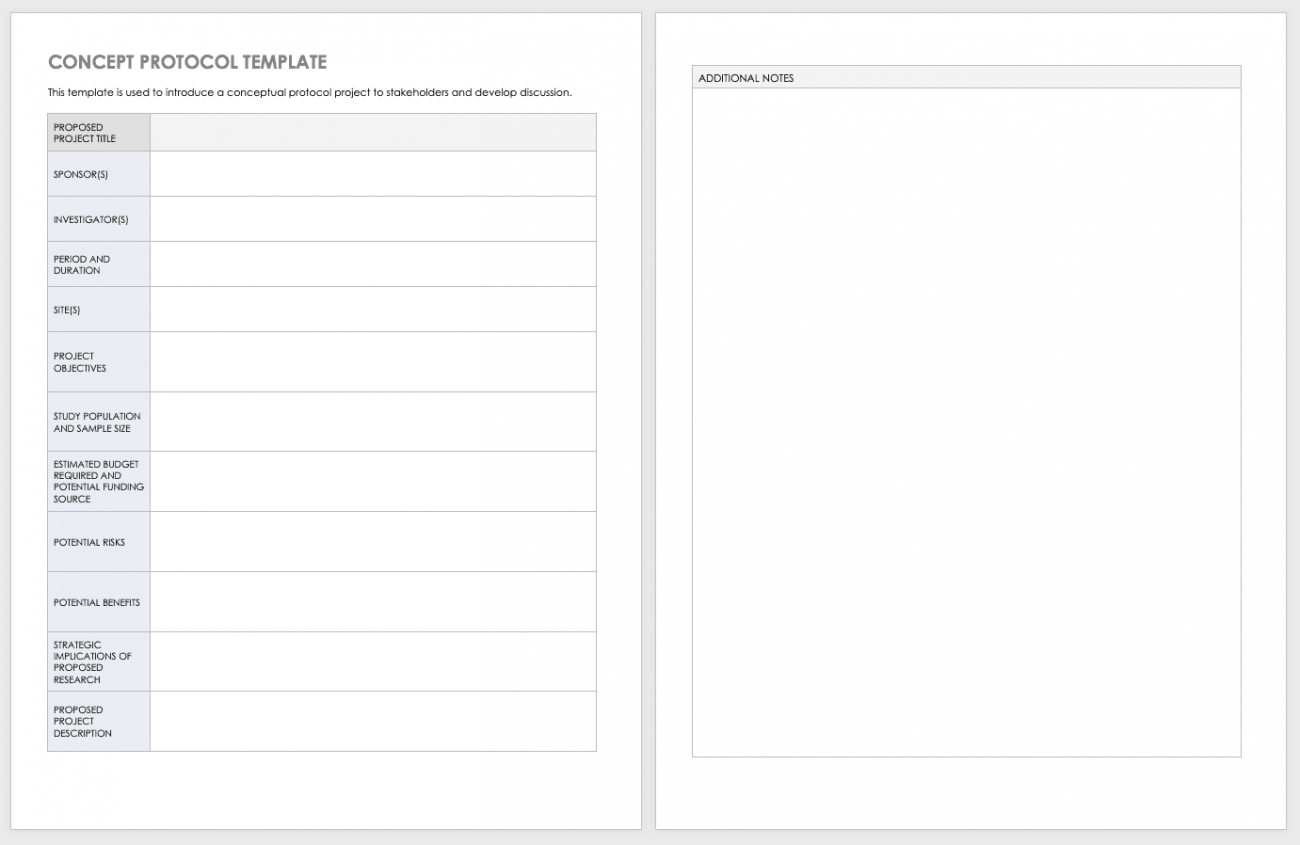
Clinical Trial Protocol Template - This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. This preface before finalizing and distributing the clinical trial protocol. The template is designed to enable modification suitable for the 9 particular trial. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. < n umber> principal investigator: Refer to the sections below for additional details and. Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. This clinical trial protocol template is a suggested format for phase 2 and 3. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in adult patients with active psoriatic arthritis.
Refer to the sections below for additional details and. < n umber> principal investigator: This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in adult patients with active psoriatic arthritis. This clinical trial protocol template is a suggested format for phase 2 and 3. The template is designed to enable modification suitable for the 9 particular trial. This preface before finalizing and distributing the clinical trial protocol. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure.
The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. This preface before finalizing and distributing the clinical trial protocol. This clinical trial protocol template is a suggested format for phase 2 and 3. < n umber> principal investigator: The template is designed to enable modification suitable for the 9 particular trial. Refer to the sections below for additional details and. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in adult patients with active psoriatic arthritis. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated.
Phase 1 Clinical Trial Protocol Template
This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. < n umber> principal investigator: A protocol template for a phase iv clinical trial of secukinumab 300 mg and.
Free Clinical Trial Templates Smartsheet
This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. Refer to the sections below for additional details and. Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. This clinical trial protocol template is a.
Free Clinical Trial Templates Smartsheet
The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in adult patients.
Phase 1 Clinical Trial Protocol Template
Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in.
Clinical Trial Protocol Synopsis Template
< n umber> principal investigator: This preface before finalizing and distributing the clinical trial protocol. The template is designed to enable modification suitable for the 9 particular trial. Refer to the sections below for additional details and. Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or.
Free Clinical Trial Templates Smartsheet
Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. This clinical trial protocol template is a suggested format for phase 2 and 3. Refer to the sections below for additional details and. < n umber> principal investigator: The template is designed to enable modification suitable for.
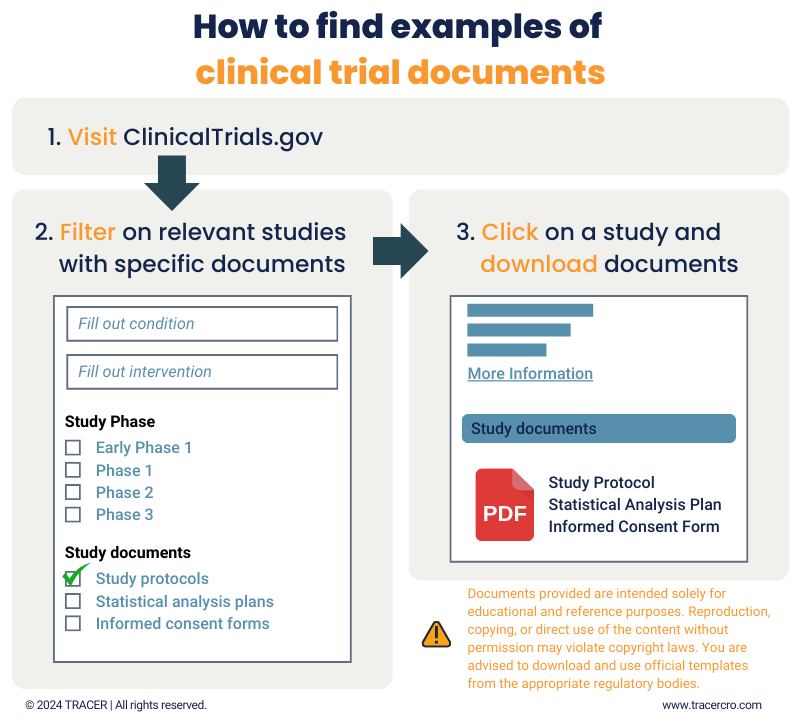
Clinical trial protocol template and example to download TRACER
< n umber> principal investigator: This preface before finalizing and distributing the clinical trial protocol. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in adult patients with active psoriatic arthritis. The template is designed to enable modification suitable for the 9 particular trial. Nih provides templates with instructional and sample text to.
Clinical Study Protocol Template
Refer to the sections below for additional details and. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. This clinical trial protocol template is a suggested format for phase 2 and 3. This preface before finalizing and distributing the clinical trial protocol. Nih provides templates with instructional and.
Free Clinical Trial Protocol Template to Edit Online
This clinical trial protocol template is a suggested format for phase 2 and 3. The template is designed to enable modification suitable for the 9 particular trial. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. < n umber> principal investigator: Refer to the sections below for additional details.
Clinical Study Protocol (CSP) Template Clinical Study Templates
Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. < n umber> principal investigator: The template is designed to enable modification suitable for the 9 particular trial. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the.
This Preface Before Finalizing And Distributing The Clinical Trial Protocol.
Refer to the sections below for additional details and. Nih provides templates with instructional and sample text to help write clinical protocols for phase 2 or 3 trials with ind/ide applications or. A protocol template for a phase iv clinical trial of secukinumab 300 mg and 150 mg in adult patients with active psoriatic arthritis. The template is designed to enable modification suitable for the 9 particular trial.
This Protocol Template Is A Tool To Facilitate The Development Of A Research Study Protocol Specifically Designed For The Investigator Initiated.
< n umber> principal investigator: The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. This clinical trial protocol template is a suggested format for phase 2 and 3.