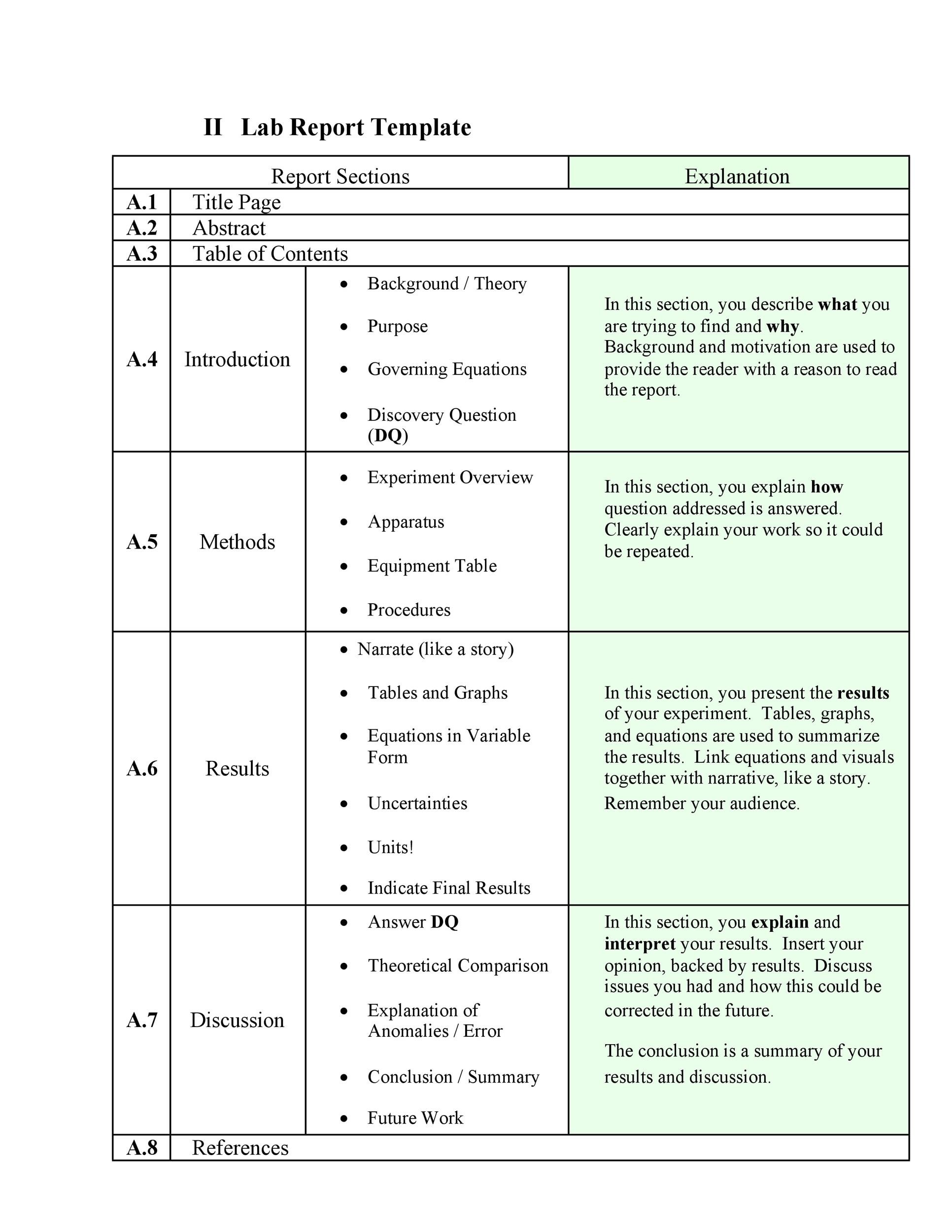
Clinical Study Report Template - This document provides recommendations for the structure and content of clinical study reports submitted to the fda. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. Learn how to use the. The template includes sections on study design,. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. A sample template for reporting the results of a clinical study involving a drug or a device.
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. Learn how to use the. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. A sample template for reporting the results of a clinical study involving a drug or a device. The template includes sections on study design,.
The template includes sections on study design,. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. Learn how to use the. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. A sample template for reporting the results of a clinical study involving a drug or a device. This document provides recommendations for the structure and content of clinical study reports submitted to the fda.
CASE STUDY REPORT FORMAT GUIDELINE
This document provides recommendations for the structure and content of clinical study reports submitted to the fda. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. A sample template for reporting the results of a clinical study involving a drug or a device. Download a free template.
Free Clinical Trial Templates Smartsheet
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. A sample template for reporting the results of a clinical study involving a drug or a device. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. This.
Clinical Trial Report Template
Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. A sample template for reporting the results of a clinical study involving a drug or a device. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. The template includes sections on study design,. This.
Clinical Evaluation Report Template
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. Learn how to use the. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. The template includes sections on study design,. Download a free template for clinical study reports following ich e3.
Monitoring Report Template Clinical Trials
Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. A sample template for reporting the results of a clinical study involving a drug or a device. Learn how to use the. This.
Case Report Form Template Clinical Trials Free Design Template for
Learn how to use the. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. A sample template for reporting the results of a clinical study involving a drug or a device. The template includes sections on study design,. Find free and adaptable templates and tools for various aspects of.
Monitoring Report Template Clinical Trials Best Template Ideas
The template includes sections on study design,. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports.
Clinical Study Report RIAT Support Center
A sample template for reporting the results of a clinical study involving a drug or a device. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. Learn how to use the. This.
Clinical Summary Template Fill Online, Printable, Fillable, Blank
This document provides recommendations for the structure and content of clinical study reports submitted to the fda. Learn how to use the. The template includes sections on study design,. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols,.
Scientific Report Template
Download a free template for clinical study reports following ich e3 guidelines and fda recommendations. Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. Learn how to use the. A sample template for reporting the results of a clinical study involving a drug or a device. The.
Learn How To Use The.
Find free and adaptable templates and tools for various aspects of clinical research, such as protocols, consent forms, logs, budgets, and more. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of pharmaceuticals. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. Download a free template for clinical study reports following ich e3 guidelines and fda recommendations.
A Sample Template For Reporting The Results Of A Clinical Study Involving A Drug Or A Device.
The template includes sections on study design,.