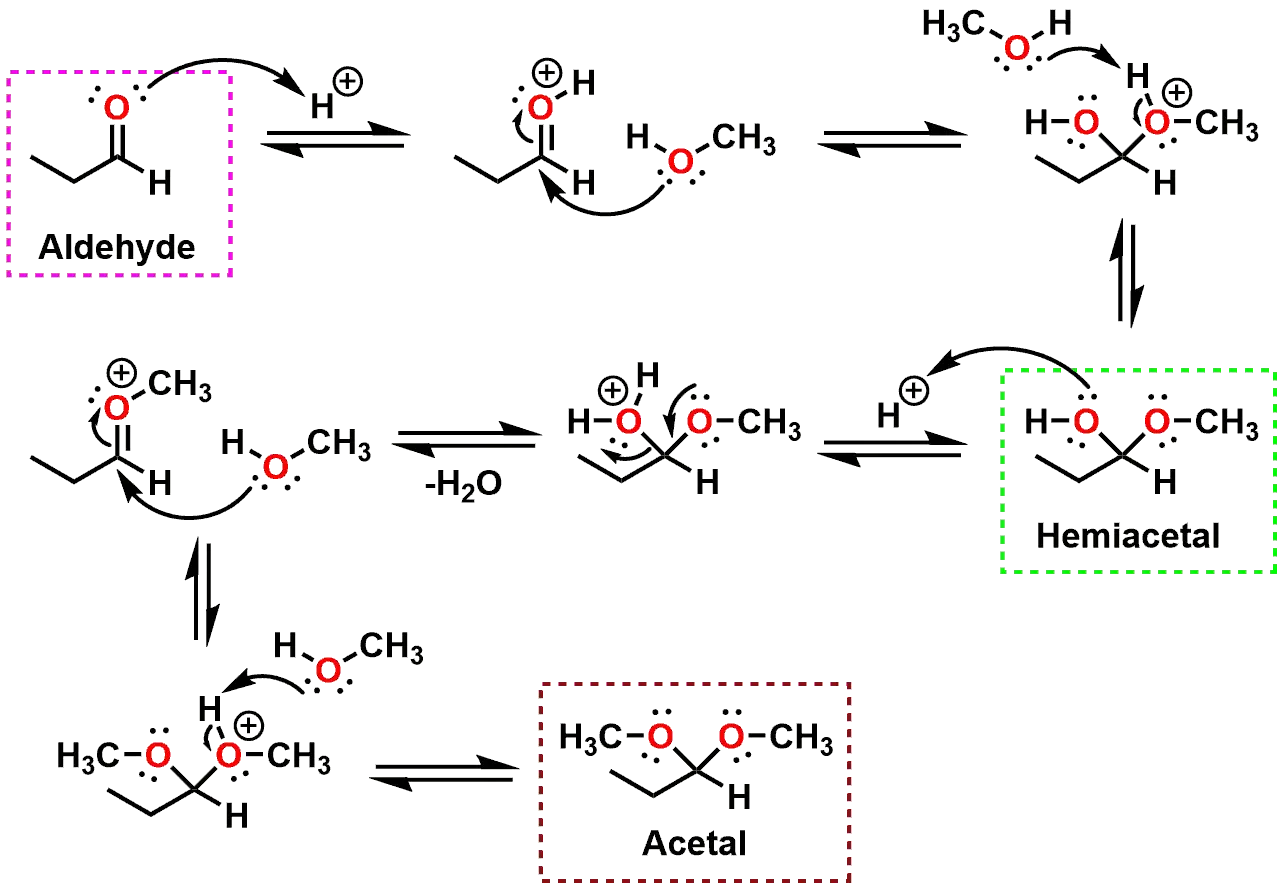
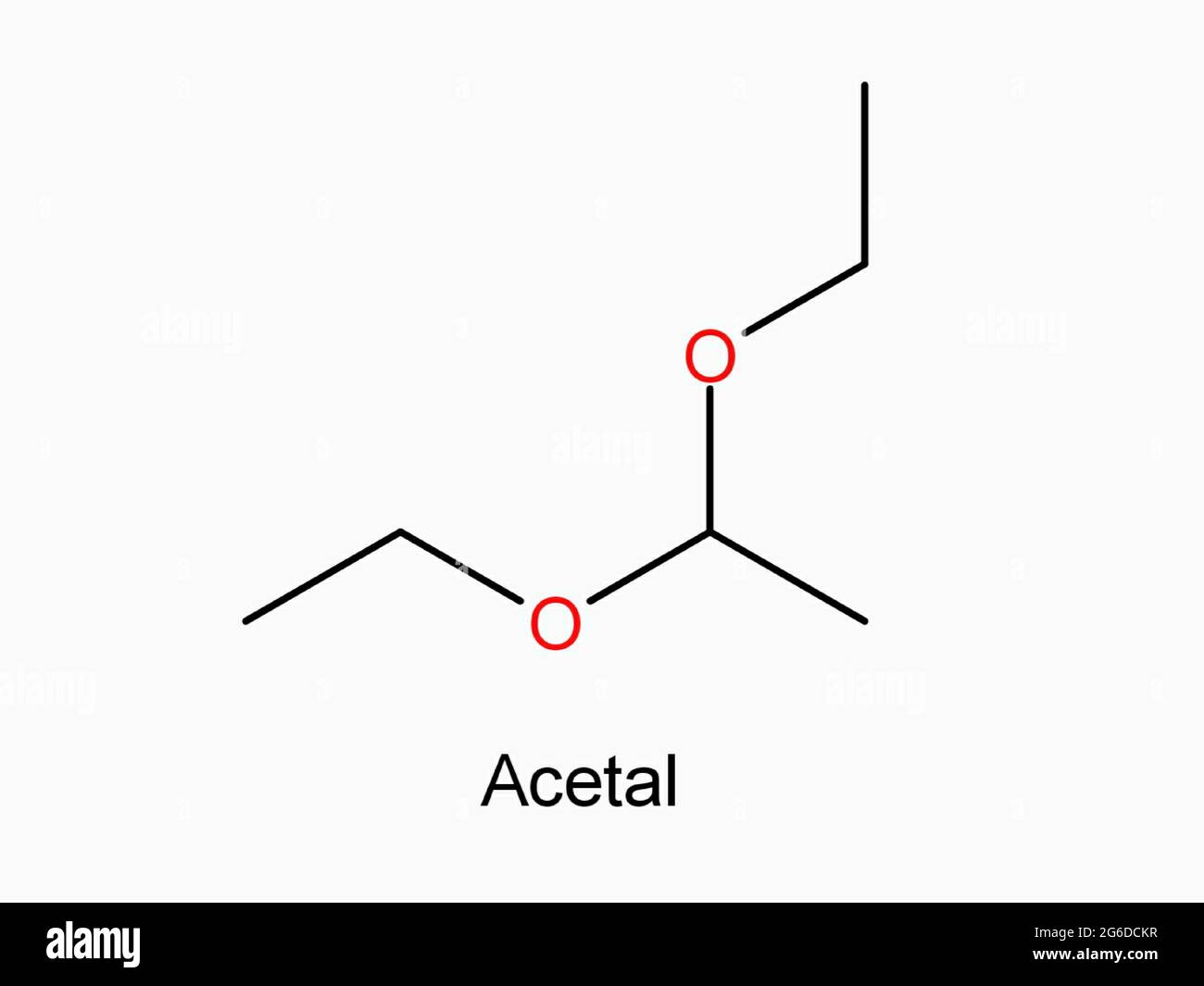
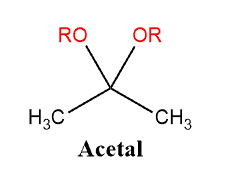
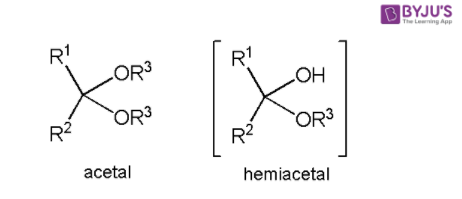
Acetal Copolymer Sheet - Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result. The carbocation that is produced is. Cacl 2 serves as a drying agent. A few grades of acetal have. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde.
The carbocation that is produced is. Cacl 2 serves as a drying agent. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. A few grades of acetal have. Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result.
Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. A few grades of acetal have. The carbocation that is produced is. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. Cacl 2 serves as a drying agent. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result.
Acetal Chemistry LibreTexts
Cacl 2 serves as a drying agent. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. The carbocation that is produced is. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. A few grades of acetal have.
Acetal Chemistry LibreTexts
A few grades of acetal have. The carbocation that is produced is. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. Formation of an acetal.
Acetal Chemistry LibreTexts
Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result. Cacl 2 serves as a drying agent. Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic.
Acetal Group 14.4 Hemiacetals And Acetals Ppt Download
From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. The carbocation that is produced is. A few grades of acetal have. Acetal's production and use as a synthetic flavoring ingredient,.
10.4 Acetals and Ketals Chemistry LibreTexts
From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. Cacl 2 serves as a drying agent. Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. A few grades of acetal have. Acetal's production and use as a synthetic flavoring ingredient, a solvent,.
Acetal Acetal Formation, Structure, Hemiacetal with Examples of Acetal
The carbocation that is produced is. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. Acetal (polyoxymethylene or pom) is a high strength, low friction engineering plastic that has excellent wear properties in both wet and dry. A few grades of acetal have. Formation of an acetal occurs when the hydroxyl group.
Acetal Definition in Chemistry
Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. The carbocation that is produced is. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and.
Acetals, Hemiacetals, Ketals & Hemiketals Organic Chemistry Explained!
The carbocation that is produced is. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. A few grades of acetal have. Formation of an acetal occurs when the hydroxyl group.
Acétal Banque de photographies et d’images à haute résolution Alamy
Cacl 2 serves as a drying agent. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result. A few grades of acetal have. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. Formation of an acetal occurs when the hydroxyl.
Acetal Group
Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. Acetal's production and use as a synthetic flavoring ingredient, a solvent, a hypnotic, in organic synthesis, perfumes, and in cosmetics may result. From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. The carbocation.
Acetal (Polyoxymethylene Or Pom) Is A High Strength, Low Friction Engineering Plastic That Has Excellent Wear Properties In Both Wet And Dry.
From organic syntheses, here’s a link to a classic procedure for making the diethyl acetal of acetaldehyde. The carbocation that is produced is. A few grades of acetal have. Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water.
Acetal's Production And Use As A Synthetic Flavoring Ingredient, A Solvent, A Hypnotic, In Organic Synthesis, Perfumes, And In Cosmetics May Result.
Cacl 2 serves as a drying agent.
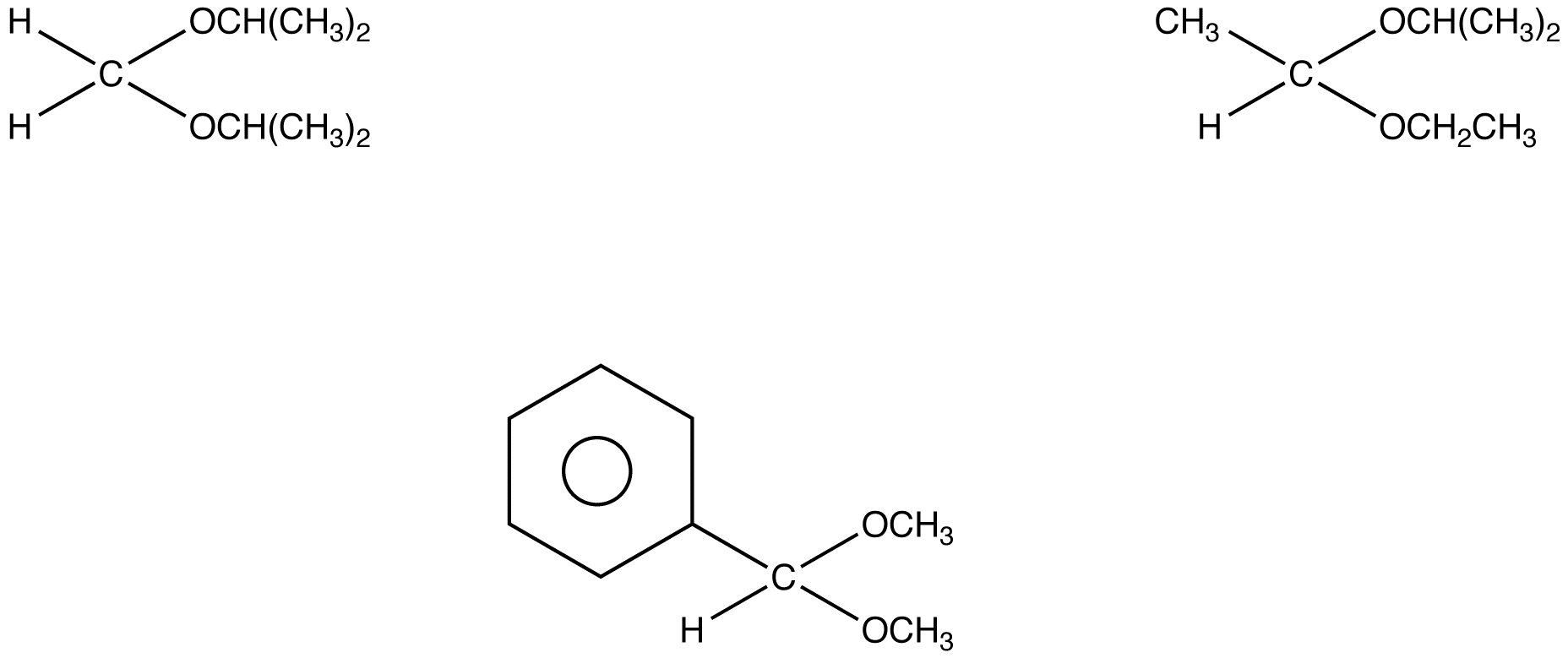
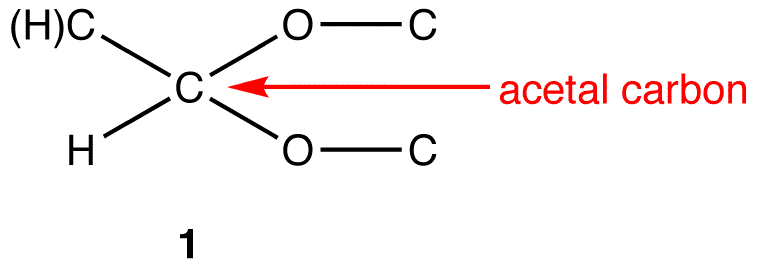
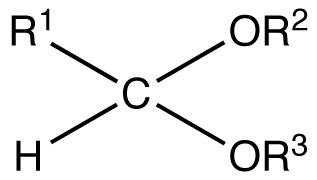
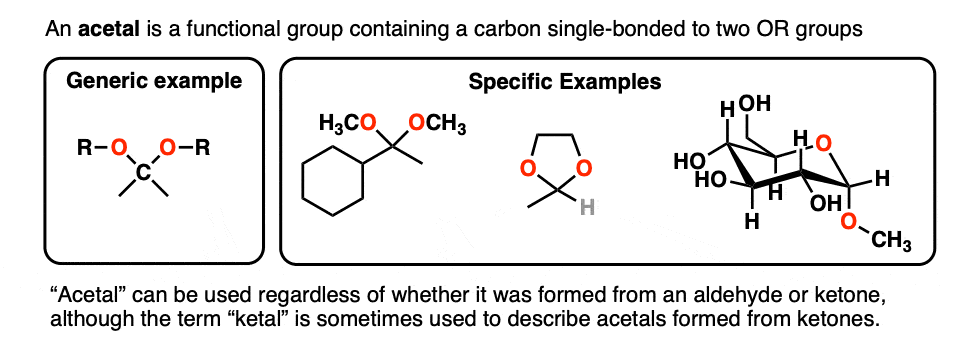
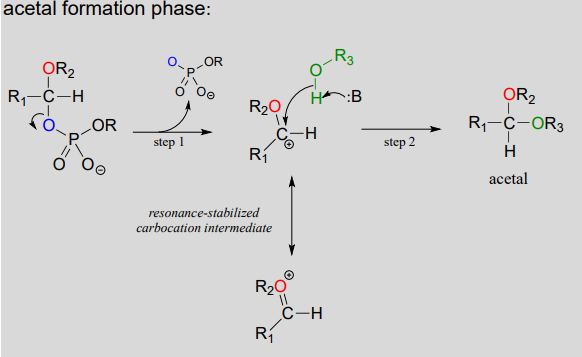
:max_bytes(150000):strip_icc()/Acetal_general_structure-f66ec08bae954edd8186a2498bd7edb5.png)