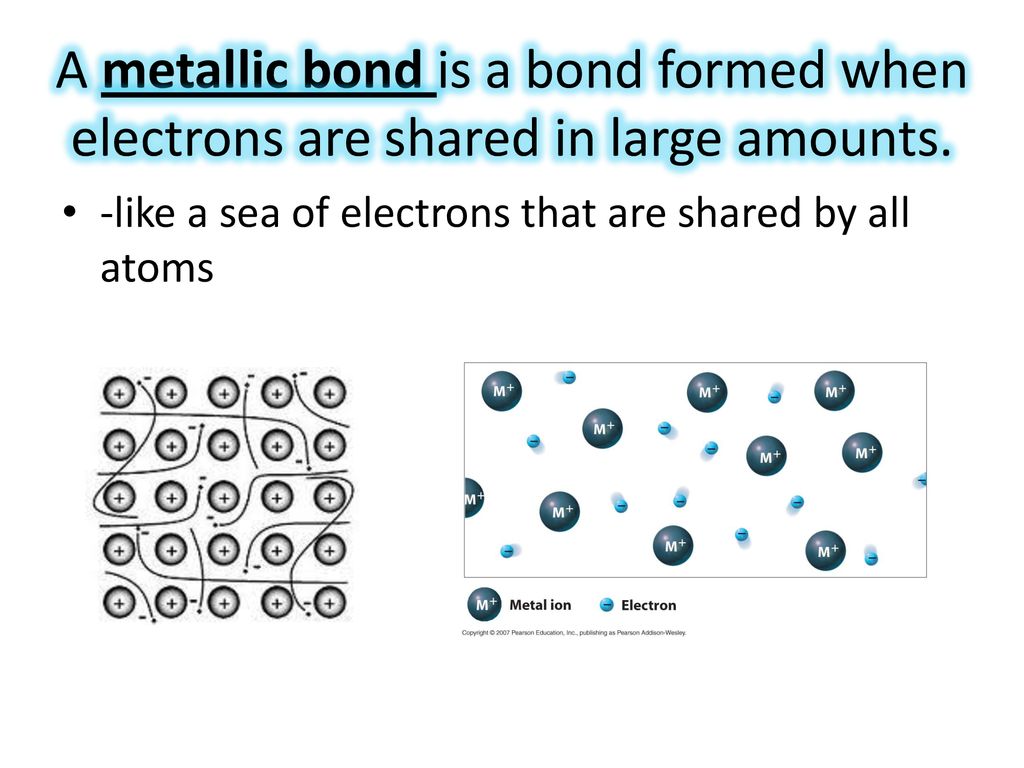
A Bond Formed From Shared Electrons - A bond formed between shared electrons. You can think of a polar covalent bond as being partway between a. Which of the following describes hydrogen bonding? Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. Interactions between positive and negative regions of polar. A polar bond is a covalent bond in which the electrons are shared unequally. A bond formed between donated electrons. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would.
Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. A polar bond is a covalent bond in which the electrons are shared unequally. A bond formed between shared electrons. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would. Interactions between positive and negative regions of polar. Which of the following describes hydrogen bonding? A bond formed between donated electrons. You can think of a polar covalent bond as being partway between a.
You can think of a polar covalent bond as being partway between a. A polar bond is a covalent bond in which the electrons are shared unequally. A bond formed between donated electrons. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would. Interactions between positive and negative regions of polar. A bond formed between shared electrons. Which of the following describes hydrogen bonding? Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from.
Covalent Bonding. Covalent Bond Chemical bond formed by the sharing of
A bond formed between shared electrons. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would. Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. You can think of a polar.
CHEMISTRY PHYSIO ppt download
Interactions between positive and negative regions of polar. A bond formed between donated electrons. A polar bond is a covalent bond in which the electrons are shared unequally. You can think of a polar covalent bond as being partway between a. A bond formed between shared electrons.
Chemical Bonding. ppt download
A bond formed between donated electrons. Interactions between positive and negative regions of polar. You can think of a polar covalent bond as being partway between a. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would. Which of the following describes hydrogen bonding?
Covalent Bonding (Biology) — Definition & Role Expii
Which of the following describes hydrogen bonding? Interactions between positive and negative regions of polar. A bond formed between donated electrons. Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. When two atoms are bonded together via electrons, and one of those atoms has a greater.
Basic Chemistry for Earth Science ppt download
A polar bond is a covalent bond in which the electrons are shared unequally. Interactions between positive and negative regions of polar. A bond formed between donated electrons. Which of the following describes hydrogen bonding? When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would.
Structure of Atoms. ppt download
A bond formed between shared electrons. Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. Which of the following describes hydrogen bonding? Interactions between positive and negative regions of polar. A bond formed between donated electrons.
Biochemistry. ppt download
A polar bond is a covalent bond in which the electrons are shared unequally. Interactions between positive and negative regions of polar. Which of the following describes hydrogen bonding? A bond formed between shared electrons. You can think of a polar covalent bond as being partway between a.
Covalent Bonding Two Day Lesson. ppt download
You can think of a polar covalent bond as being partway between a. Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed.
Day 2 The Nature of Matter Notes ppt download
Which of the following describes hydrogen bonding? A bond formed between shared electrons. A polar bond is a covalent bond in which the electrons are shared unequally. Interactions between positive and negative regions of polar. You can think of a polar covalent bond as being partway between a.
Chapter 2 Atoms and Bonding ppt download
Which of the following describes hydrogen bonding? Interactions between positive and negative regions of polar. A bond formed between shared electrons. You can think of a polar covalent bond as being partway between a. A bond formed between donated electrons.
You Can Think Of A Polar Covalent Bond As Being Partway Between A.
A bond formed between donated electrons. Oxygen atoms have a stronger pull on the electrons shared within a covalent bond formed between oxygen and hydrogen protons are transferred from. When two atoms are bonded together via electrons, and one of those atoms has a greater pull upon the electrons shared, the bond formed would. Which of the following describes hydrogen bonding?
A Polar Bond Is A Covalent Bond In Which The Electrons Are Shared Unequally.
A bond formed between shared electrons. Interactions between positive and negative regions of polar.