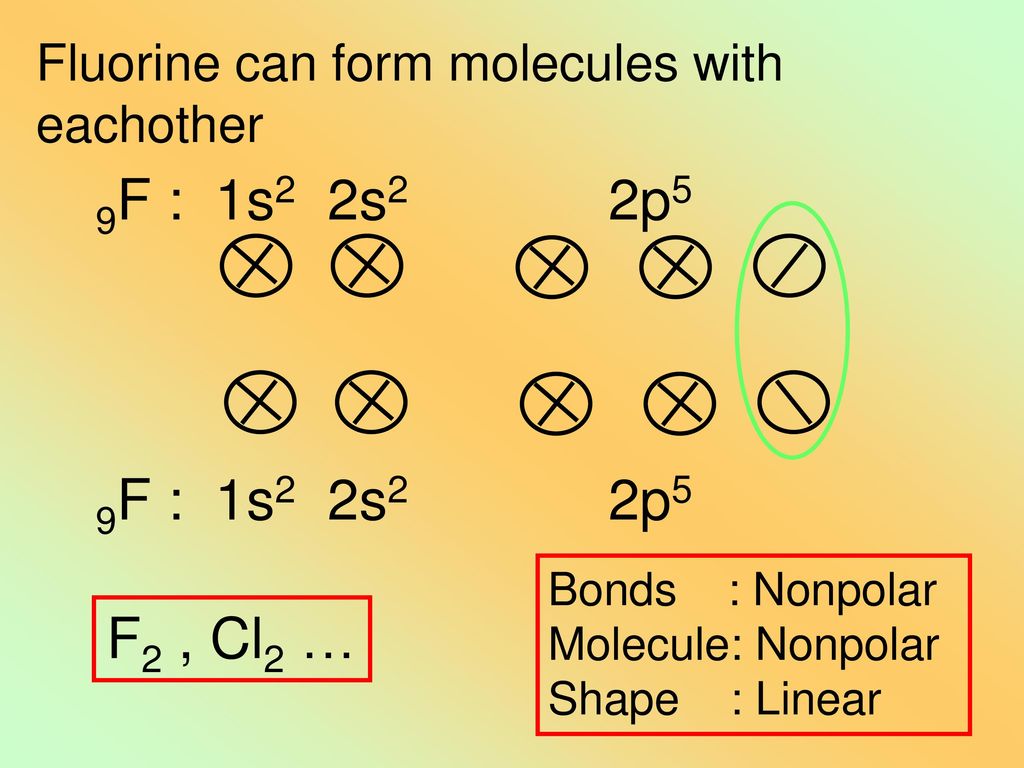
Fluorine Can Form ____ Bonds With Other Atoms. - This makes the fluoride anion so formed unstable. What is the nature of the reaction of attack of fluorine gas on aluminium metal? Is it spontaneous in nature? It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine. What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. As fluorine is the most electronegative element in the periodic table, it. I have studied reactions of. Why is fluorine the most electronegative atom?
It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine. As fluorine is the most electronegative element in the periodic table, it. I have studied reactions of. Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. Why is fluorine the most electronegative atom? This makes the fluoride anion so formed unstable. What is the nature of the reaction of attack of fluorine gas on aluminium metal? Is it spontaneous in nature?
Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. Why is fluorine the most electronegative atom? This makes the fluoride anion so formed unstable. I have studied reactions of. It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine. As fluorine is the most electronegative element in the periodic table, it. Is it spontaneous in nature? What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. What is the nature of the reaction of attack of fluorine gas on aluminium metal?
Chapter 6 Chemical Bonding. ppt download
Is it spontaneous in nature? As fluorine is the most electronegative element in the periodic table, it. This makes the fluoride anion so formed unstable. Why is fluorine the most electronegative atom? Fluorine, though higher than chlorine in the periodic table, has a very small atomic size.
Lewis Structures. ppt download
This makes the fluoride anion so formed unstable. What is the nature of the reaction of attack of fluorine gas on aluminium metal? What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. I have studied reactions of. Fluorine, though higher than chlorine in the periodic table, has a very small atomic size.
Lewis Dot Structures. ppt download
What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. This makes the fluoride anion so formed unstable. Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine. What is the nature of the reaction of attack of fluorine.
Introduction to Matter Measuring Matter Changes in Matter Elements and
Is it spontaneous in nature? This makes the fluoride anion so formed unstable. I have studied reactions of. As fluorine is the most electronegative element in the periodic table, it. It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine.
Covalent Bonding And Molecular Compounds. ppt download
This makes the fluoride anion so formed unstable. Why is fluorine the most electronegative atom? I have studied reactions of. As fluorine is the most electronegative element in the periodic table, it. What is the nature of the reaction of attack of fluorine gas on aluminium metal?
CHEMICAL BONDS. ppt download
I have studied reactions of. Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. As fluorine is the most electronegative element in the periodic table, it. It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine. Why is fluorine the most electronegative atom?
CHEMICAL BONDS. ppt download
Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. What is the nature of the reaction of attack of fluorine gas on aluminium metal? Why is fluorine the most electronegative atom? As fluorine is the most electronegative element in the periodic table, it. This makes the fluoride anion so formed unstable.
Bonds…. Covalent Bonds Chapter 5 sec. 3 Mr. Sapalicio ppt download
As fluorine is the most electronegative element in the periodic table, it. What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. This makes the fluoride anion so formed unstable. Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. It seems related to the atomic size but hydrogen has a smaller atomic size than.
Lewis Dot Structures. ppt download
What is the nature of the reaction of attack of fluorine gas on aluminium metal? I have studied reactions of. This makes the fluoride anion so formed unstable. Why is fluorine the most electronegative atom? It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine.
Fluorine Electron Configuration With Full Orbital Diagram
Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. I have studied reactions of. Why is fluorine the most electronegative atom? What is the nature of the reaction of attack of fluorine gas on aluminium metal? Is it spontaneous in nature?
As Fluorine Is The Most Electronegative Element In The Periodic Table, It.
Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. Why is fluorine the most electronegative atom? This makes the fluoride anion so formed unstable. What is the nature of the reaction of attack of fluorine gas on aluminium metal?
Is It Spontaneous In Nature?
What should be the oxidation state of $\\ce{f}$ in $\\ce{hof}$. It seems related to the atomic size but hydrogen has a smaller atomic size than fluorine. I have studied reactions of.